Access the full version of this brief with detailed access to the complete recap of over 300 global updates in the last month, as well as a curated database of +7,000 regulation updates, market reports and science papers.
February 2026 was defined by cross-border consolidation on an unprecedented scale. Organigram acquired Sanity Group for €227M — Europe’s largest cannabis deal — as Germany crossed 201 tonnes imported and €1 billion in pharmacy sales. In Australia, the LGP–Cannatrek merger moved toward A$112M in combined revenue as Montu announced fully-year unaudited revenues of A$385M. Capital is flowing into maturing medical markets faster than regulation can keep pace.
Yet the institutional response remained uneven. Germany’s MedCanG amendment deadlocked over telemedicine and mail-order restrictions. Brazil’s RDC 1,015 authorised domestic cultivation but confined it to CBD-based pharmaceuticals. North Macedonia seized 40 tonnes from licensed producers and revoked 18 licences. Record deal flow and record volumes launched in parallel with political reversals, integrity crackdowns, and access bottlenecks far from resolved. Key figures from February 2026 include:
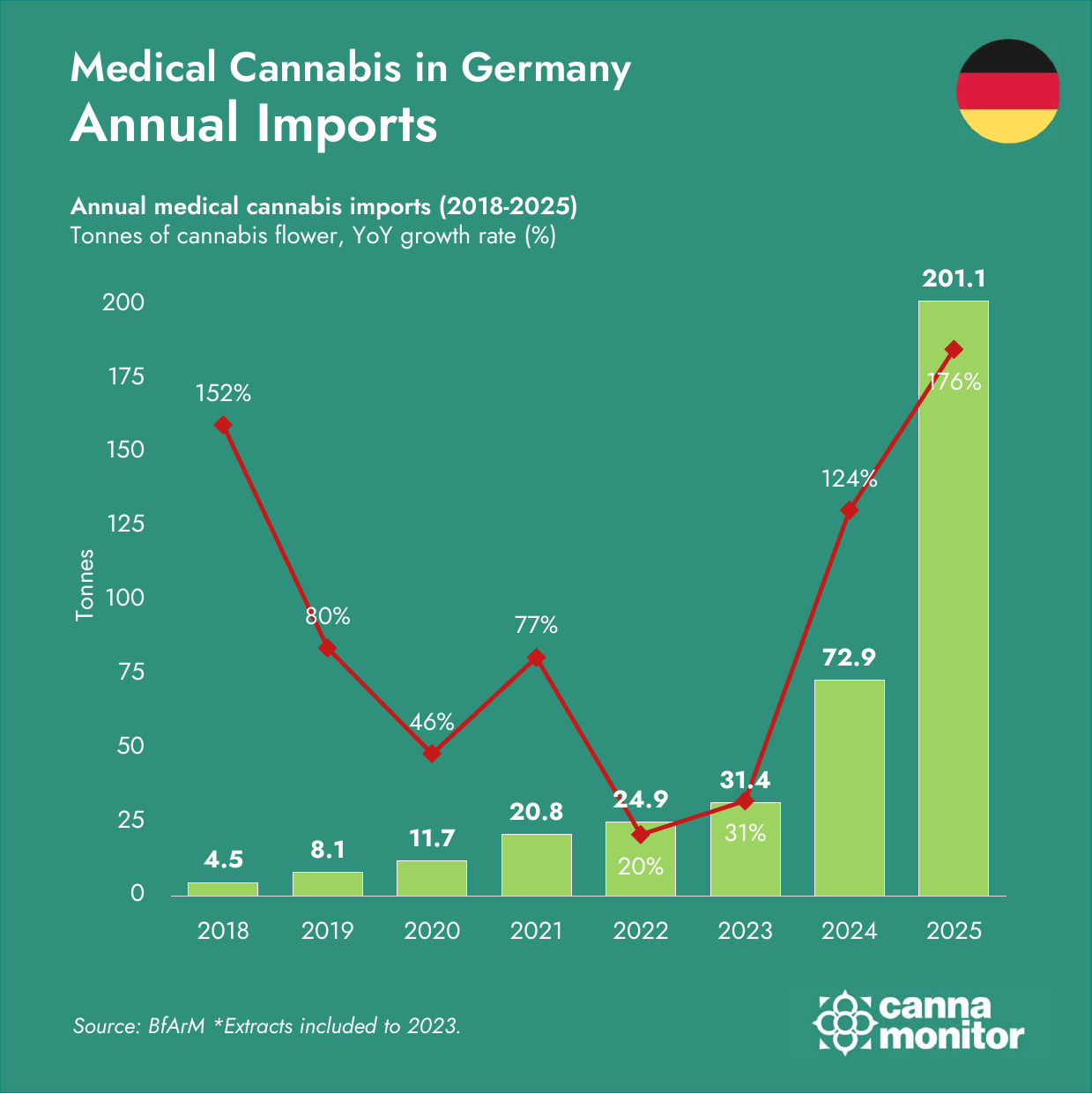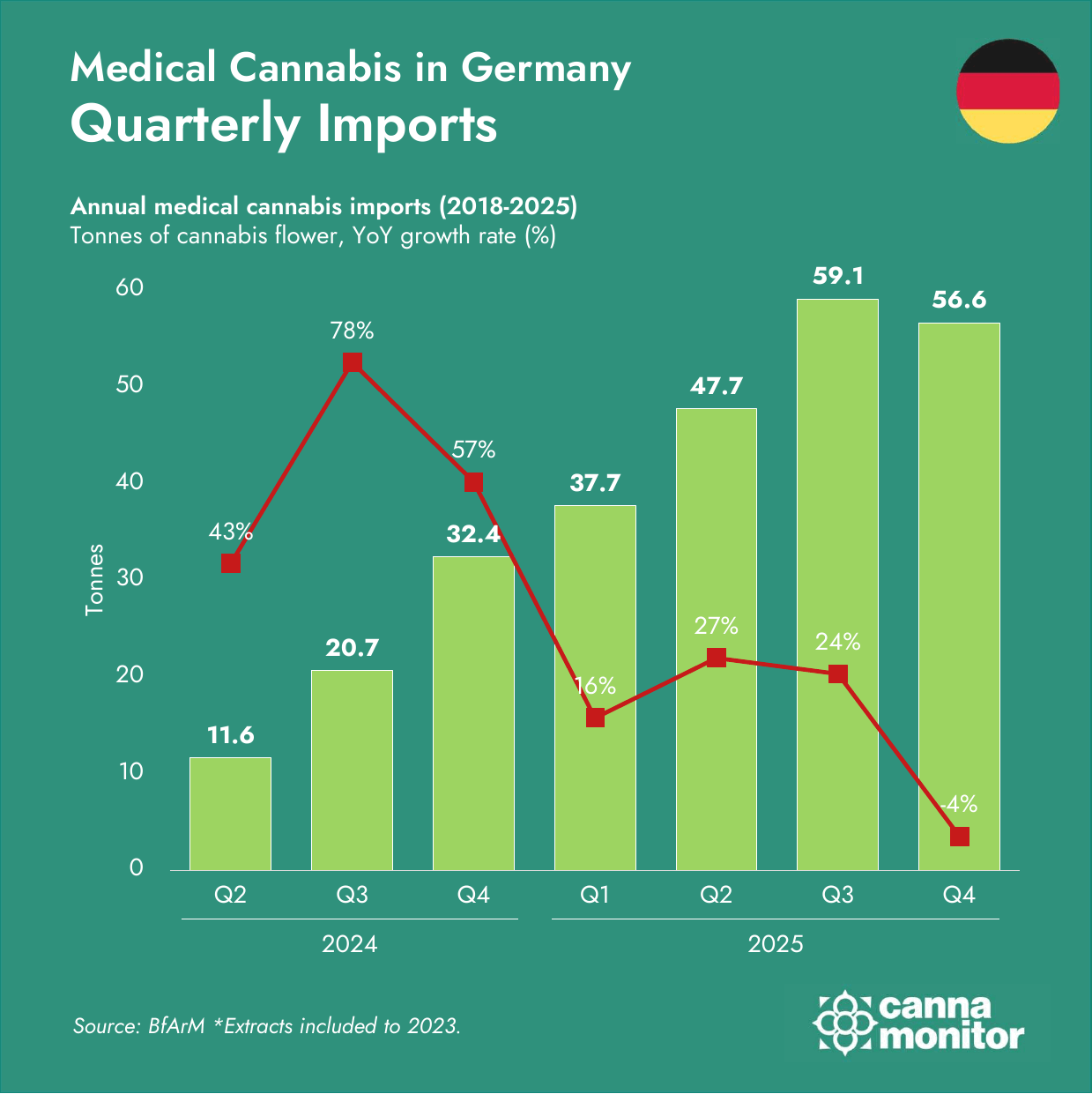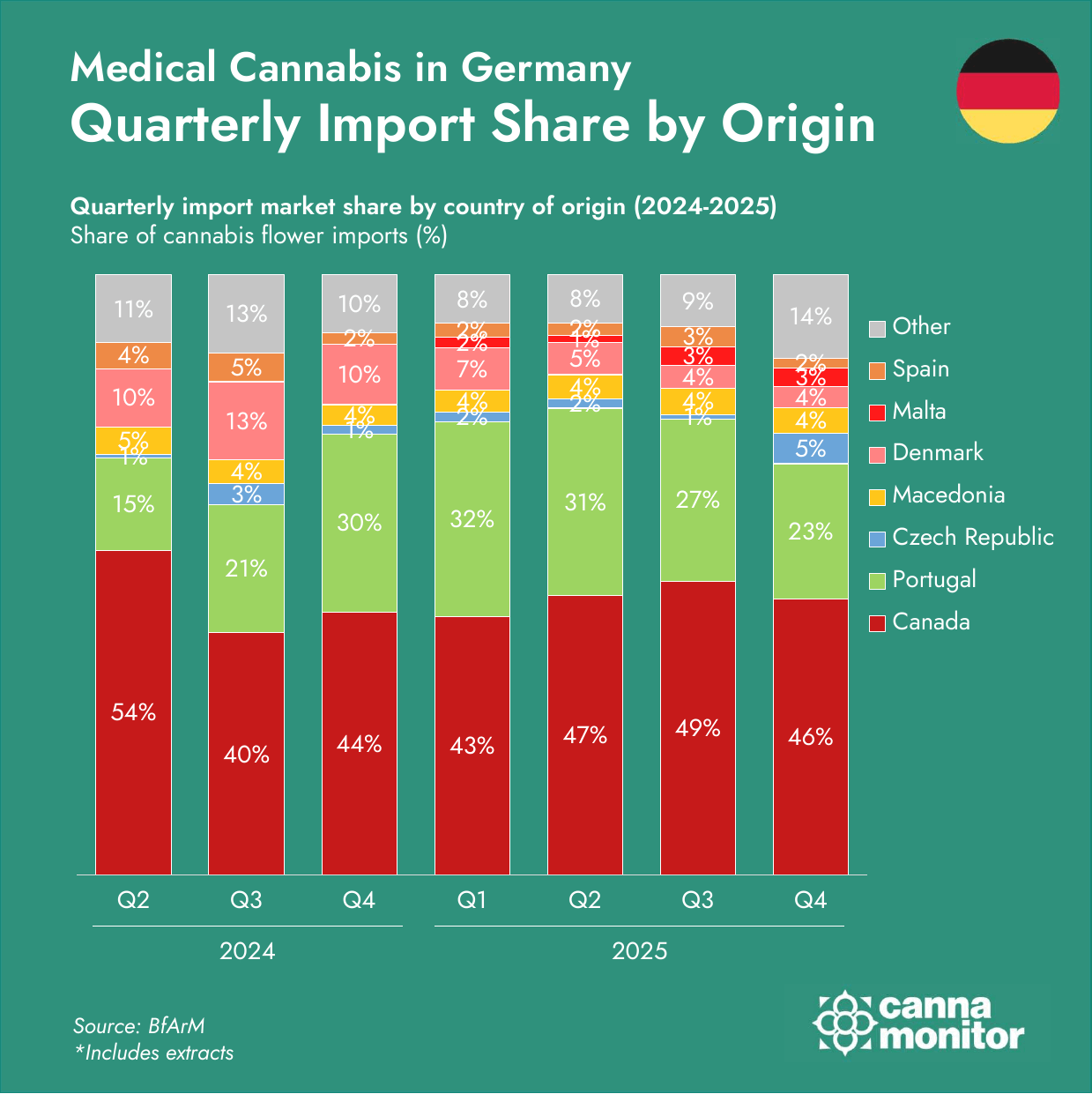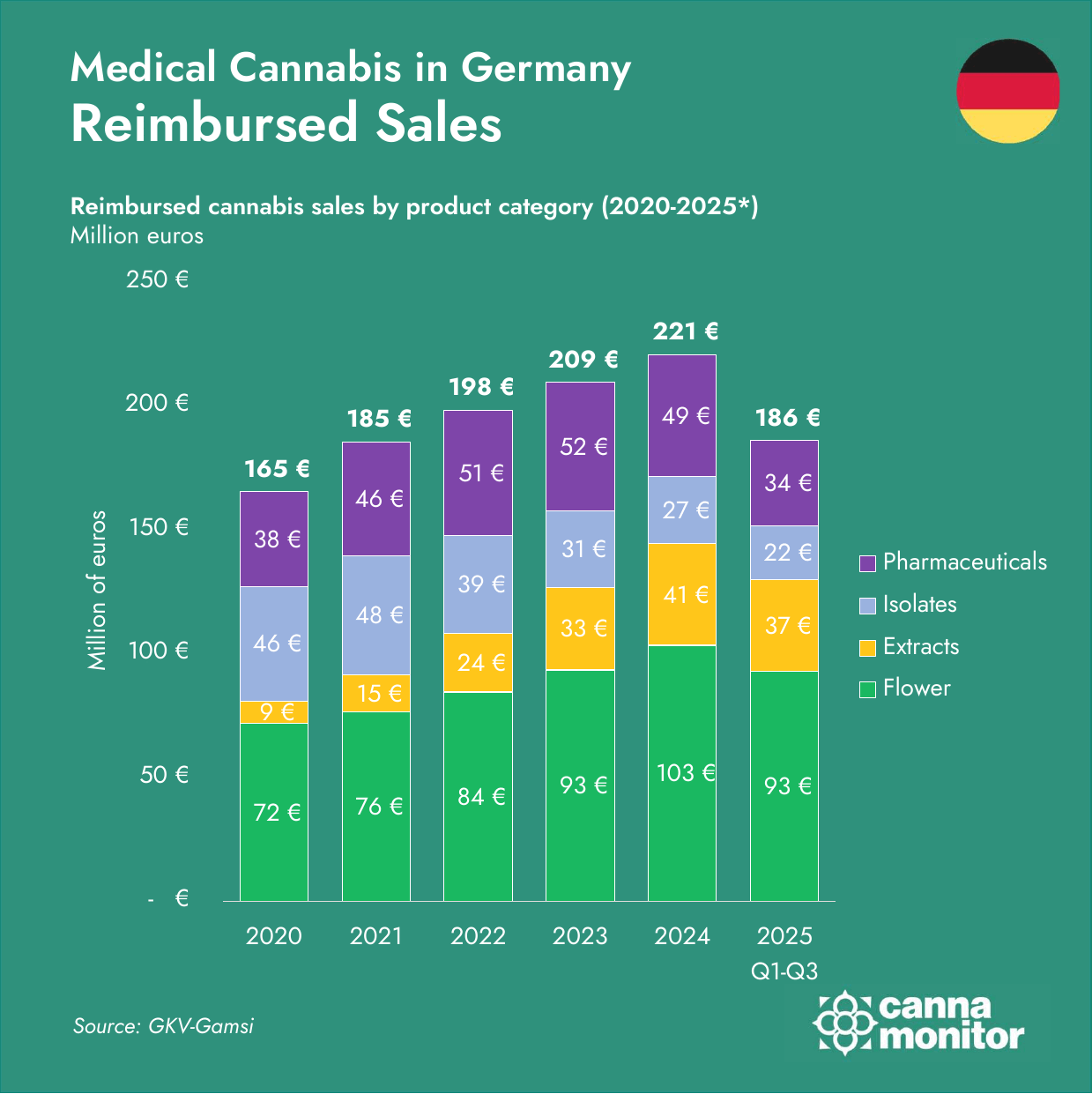
Germany reaches 201 tonnes imported in 2025 (+176% YoY)
Organigram acquires Sanity for €227M — one of Europe’s largest M&A
Federal cuts threaten 565 active NIH cannabis experiments worth $310M+
January retail sales in the USA reach $2.07B across 15 states (+4.6% YoY)
Canada sales record in December of C$503.7M; full-year C$5.62B (+4.1%)
An estimated ~900,000 Brazilian patients already use cannabis extracts
Australian merger of LGP–Cannatrek targets A$112M combined revenue
UK regulators included 11,456 CBD products on the FSA novel food register
Just 70 of ~10,000 Swiss patients are publicly reimbursed
Global Epidiolex sales cross $1.1B in 2025 (+9%)
Feel free to contact us at info@cannamonitor.com with any suggestions.
Table of Contents
New Market Intelligence Webinar:
Germany: 2026 Market & M&A Trends
Following the Money into Europe’s Biggest Cannabis Market
🏛️
Regulation
Germany MedCanG amendment deadlocked as US rescheduling stalls. Brazil’s RDC 1,015 restricts medical cannabis to CBD-based pharmaceuticals with mandatory drug registration. North Macedonia seizes 40 tonnes and revokes 18 of 60 licences. UK Crown Court clarifies statutory defence for 90,000+ patients. In Australia, 790 TGA submissions find SAS framework “not fit-for-purpose”. South Africa gazettes 750g possession cap and five-plant limit. Swiss Grashaus pilot halves illegal-market use; Dutch 68-seat majority secures the Wietexperiment but no post-evaluation budget.
Market
Organigram acquires Sanity Group for €227M in one of Europe’s largest deals — as Germany crosses 201 tonnes imported and €1bn in pharmacy sales. Top-4 US MSOs post $300M+ quarters with Curaleaf closing a record $500M notes offering. In Canada, record C$5.62bn retail year but two LP groups enter CCAA. LGP–Cannatrek merger creates an A$112M Australian champion as pastilles reach 30% of SAS-B prescriptions. In the Netherlands, all 10 Wietexperiment growers are now active.
Science
Dutch study finds that eliminating the vegetative phase boosts annualised yield 12% while cutting energy use 30% and labour 37%, with Grade A flower rising from 20% to 35%.
Infographic of the month:
Medical Cannabis Imports to Germany
🇩🇪 Germany — Over 200 tonnes imported in 2025 as Organigram Acquires Sanity Group
🔭On the radar: BGH ruling on Bloomwell/Wellster telemedicine legality — expected end of March 2026.
🏛️ Regulation — MedCanG Stalls As Coalition Splits On Mail-Order Ban
Germany’s medical cannabis reform stalled as the coalition fractured over the proposed mail-order ban. The SPD rejected blanket restrictions and the Greens sought to remove the shipping ban entirely, leaving the MedCanG amendment deadlocked. Meanwhile, recreational cannabis clubs met less than 0.1% of demand, and sub-national enforcement continued to fragment across the Länder.
- The coalition split on the mail-order ban deepened as SPD health spokesperson Matthias Mieves rejected blanket bans on telemedicine prescribing, warning they would harm rural and mobility-limited patients. The Greens’ Linda Heitmann confirmed the reform is stalled and the coalition is deadlocked, while an EcoCan evaluation showed declining youth cannabis consumption since legalisation.
- A 60,000-signature petition reached the Bundestag Petitions Committee on 23 February, arguing that in-person-only prescribing and a mail-order ban create a two-tier healthcare system. The underlying Petition 184070, filed in October 2025, cited up to €100 per in-person visit and warned of rural access collapse. The CDU’s health-protection narrative faced critique: offences halved to 100,000 and youth use declined post-legalisation.
- The DHV election analysis found no clear cannabis policy winner ahead of Baden-Württemberg‘s 8 March 2026 state vote. Greens and SPD ranked highest; CDU and AfD explicitly opposed to liberalisation.
- The genuine-vs-non-genuine patient dispute intensified as BfArM data showed 142 tonnes imported in the first three quarters of 2025 — nearly double the full-year 2024 total — alongside stagnating GKV prescriptions. Lower Saxony‘s health minister called for mandatory in-person first prescriptions.
- The BGH opened review of whether Bloomwell’s and Wellster’s platforms constitute unlawful advertising under the Heilmittelwerbegesetz. The court flagged EU freedom of services and may refer questions to the CJEU. Decision expected end of March 2026. ⏳ Pending.
- Weed.de warned the proposed amendment would cut off access for hundreds of thousands of patients, citing 47% patient stigmatisation. The ApothekenRechtTag 2026 examined prescribing and dispensing legal risks for pharmacists amid the evolving landscape.
- The Bundesrat proposed subjecting medical cannabis to the Arzneimittelpreisverordnung (fixed-price regulation); the federal government said it will review the proposal. Health minister Nina Warken targeted e-prescription reliability, boosting Gematik‘s digital strategy.
- The pharmacy reform draft raised alarm over pricing transparency and reimbursement, with Medipolis owner Dr Christian Wegner warning of existential threats to specialised outpatient care. Dr Markus Veit questioned certified vaporiser suitability for cannabis rosin, calling for product-specific IIb risk classification.
- The CDU party conference voted to reverse recreational cannabis legalisation. The Frauen Union demanded full CanG repeal, prompting an open letter from BvCW’s Lisa Haag to chair Nina Warken. Aurora Cannabis criticised the in-person-only plan as limiting rural patient access, while Cansativa and BPC engaged MPs on stable medical supply at the conference.
- The Left Party urged national adult-use retail pilots after cannabis cultivation clubs met less than 0.1% of demand. The BLE halted Wiesbaden’s pharmacy dispensing pilot, citing lack of legal basis under KCanG — the third major city rejection after Frankfurt and Berlin.
- Schleswig-Holstein approved 11 clubs with 8 already growing; CDU state called for federal reforms. Rheinland-Pfalz enforced a root ban on cannabis cuttings, testing KCanG’s propagation material definition.
- Bavaria lifted cannabis bans in state parks after the Administrative Court’s November 2025 ruling became legally binding. A Bavarian court clarified the plant definition: a planted cutting becomes a cannabis plant under KCanG.
- Hamburg’s Bergedorf cannabis cultivation association quit despite holding a licence, citing insurmountable operational barriers under KCanG — reinforcing the pattern of club non-viability.
- German Customs reported over 10 tonnes of US cannabis seized at Cologne/Bonn airport in 2025; the EMCDDA warned of a sharp increase in transatlantic cannabis parcels, underlining persistent illicit supply pressure alongside legal market expansion.
💼 Market — Organigram Acquires Sanity Group For €227M In Europe’s Largest Cannabis Deal
Germany’s medical cannabis market crossed €2 billion as Organigram‘s €227M acquisition of Sanity Group — the largest European cannabis M&A to date — signalled a decisive Canadian capital pivot toward German market infrastructure. Record imports of 201.1 tonnes and pharmacy sales surpassing €1 billion underscored the scale, while fewer than 1% patient penetration pointed to substantial further upside.
- Organigram agreed to acquire Sanity Group for up to €227M, comprising €113.4M upfront and €113.8M in earnouts. Sanity Group grew net revenue from €9M (2023) to €60M in 2025 with a 47% gross margin, reaching estimated #2 market share. BAT invested C$65.2M in equity; shareholder vote set for 30 March 2026. ⏳ Pending.
- Albert Schwarzmeier (Enua CEO) outlined accelerating consolidation dynamics, predicting a wave of M&A after any MedCanG adjustment. Operators without vertical integration, own pharmacy networks or data capabilities face margin pressure; Enua sold two tonnes in December 2025 alone.
- ECS Botanics signed a distribution deal with Nimbus Health to supply budget OzSun flower to German pharmacies, with first sales expected in April 2026. Cannaflos partnered with Galaxia Vertical on GMP-certified flower cultivated by Pharma Rolly in North Macedonia.
- Germany imported a record 201.1 tonnes of medical cannabis in 2025 (+176% YoY) — the largest annual volume any regulated import market has recorded. Pharmacy sell-out surpassed €1 billion for the first time; a Bloomberg feature cited BPC‘s estimate of ~€2 billion including platform fees.
- Supply corridors diversified sharply beyond Canada–Portugal: Malta surged +2,900%, Czechia +261% and the United Kingdom +1,001%. Q4 registered the first-ever quarterly import decline (−4%), as pre-restriction stocking unwound and competitive dynamics shifted.
- Will Muecke noted fewer than 1% patient penetration, suggesting the market’s growth runway remains long. Search data analysis of 2,500 top German cannabis queries found demand split equally between access routes (~22%) and cultivation topics (~22%).
- A Cantourage survey found only one in ten GPs willing to prescribe medical cannabis across Germany’s 20 largest cities — zero positive responses in Berlin, Bielefeld or Frankfurt — highlighting dependency on telemedicine platforms for patient access.
- Grünhorn‘s CanDoc platform launched video consultations alongside questionnaire-based prescriptions, positioning itself as one of the most comprehensive telemedicine providers with over 130,000 registered users. The move came as the MedCanG amendment threatened to restrict digital access channels.
🔬 Science — Lancet Finds No Rise In Cannabis Use After CanG Legalisation
A landmark Lancet Regional Health – Europe difference-in-differences study by the University Medical Center Hamburg-Eppendorf found no short-term rise in cannabis use or driving under the influence after CanG legalisation, comparing Germany against 26 EU controls. The finding directly undermines the CDU’s public-health justification for reversal and aligns with declining youth consumption data since the law took effect.
- A Cochrane systematic review evaluated cannabis-based medicines for neuropathic pain in adults, synthesising the latest randomised controlled trial evidence on efficacy and tolerability — a key reference as Germany’s prescribing debate intensifies around the MedCanG amendment.
- A preclinical study found THC boosts growth factor release by cultured adipose stem cells, suggesting a novel regenerative mechanism that could broaden the therapeutic rationale beyond established indications.
- Germany’s CanG-mandated recreational research projects were reported as well planned with municipalities ready, but post-election uncertainty under the new CDU-led coalition now threatens funding continuity — raising the prospect that critical evaluation data may never be collected.
🇺🇸 United States — Rescheduling Stalls As Political Resistance Crystallises And States Fill The Federal Vacuum
🔭On the radar: Ohio hemp beverage sales ban takes effect 20 March; Florida recreational referendum ballot qualification confirmation pending.
🏛️ Regulation — GOP Senator Tells Trump Rescheduling Was “Poorly Advised” As 13 States Advance Ryan’s Law
Federal cannabis rescheduling stalled as Republican Senator Ted Budd told the president directly that the process was “poorly advised,” crystallising political resistance to Schedule III reclassification. At state level, Ryan’s Law advanced medical access in 13 states for terminally ill hospital patients, while Ohio’s referendum battle and Virginia’s adult-use legalisation dominated the adult-use frontier.
- The proposed HEMP Act would create a federal CBD pathway as the hemp industry faces regulatory crossroads, but the House Agriculture chair signalled the Farm Bill hemp THC ban delay will not get a vote, leaving producers in limbo. The New York Times‘ cannabis editorial was critiqued as “bad policy journalism” for ignoring key evidence.
- A federal judge upheld CBP‘s seizure of nearly $1M from eight licensed New Mexico cannabis operators, ruling that Schedule I classification permits forfeiture regardless of state legality — underscoring the cost of rescheduling inaction in a market with 3,000+ licensees and ~$40M/month in sales.
- Ryan’s Law bills to allow medical cannabis use in hospitals for terminally ill patients advanced in at least 13 states, with Mississippi‘s House approving its version and Hawaii committees advancing their companion.
- Idaho‘s medical cannabis ballot initiative collected 45,000+ signatures with strong voter support. Kentucky‘s programme launched with gummies now available and 18,500+ cardholders, while Nebraska advanced a bill granting doctors legal immunity for medical cannabis recommendations.
- A national coalition launched a campaign to expand qualifying conditions for women’s health in 9+ states, targeting endometriosis, PCOS and menstrual disorders.
- Ohio‘s Attorney General certified a referendum petition to block SB 56 cannabis and hemp restrictions, then separately sued nine MSOs for anti-competitive collusion against smaller operators. OHCANN cited the $2 billion market and 250,000 signatures needed by 19 March. ⏳ Pending.
- Ohio communities received $35 million in cannabis tax funds after SB 56 delayed their release. Hemp beverage producers braced for a 20 March sales ban after the governor vetoed a brewery carve-out.
- Virginia‘s HB 642 passed House Appropriations to legalise adult-use sales, and resentencing bills SB 62 / HB 26 advanced in parallel. Advocates urged the Senate to strip new criminal penalties from SB 542, while Jushi expanded $30M to double production capacity ahead of retail licensing.
- Across other states: Florida‘s Smart & Safe campaign said it had enough ballot signatures for a 2026 recreational referendum, while Maine‘s repeal petition failed to qualify for the November ballot, leaving the $244M market intact. Maine separately amended its excise tax with new exemptions for inter-facility transfers.
- New York‘s OCM approved 36 new licences and New Jersey expanded cannabis labour protections beyond NLRA coverage. Massachusetts equity barriers persist: only 15% of 690 businesses are equity-owned.
- Washington advanced home-grow bill SB 6204 allowing six plants per adult, while Chicago‘s Mayor vetoed the City Council hemp ban. Washington’s CBD legal uncertainty persists as the state revisits hemp-derived definitions.
- Oregon proposed capping edibles at 10 mg THC per serving to reduce child poisonings. CaCOA‘s white paper found California’s “attractive to children” standard is undefined — a 162-brand review found 68% compliant, 22% in a grey zone.
💼 Market — Top-4 MSOs Post $300M+ Quarters As Hemp-THC Beverages Attract $15M In Venture Capital
The US cannabis market settled into a stabilisation pattern in February, with the four largest MSOs reporting quarterly revenues above US$300M and deploying over US$700M in refinancing and new debt. Hemp-derived THC beverages attracted $15M+ in venture capital and national retail distribution deals, while state-level sales data bifurcated sharply — Massachusetts crossed $10 billion cumulative versus Nevada‘s 8.6% annual decline.
- A HashDash survey of 500+ users found stress and anxiety relief drive cannabis use: 71% consume for stress, 54.8% for anxiety, yet 60% fear cannabis may worsen anxiety.
- Curaleaf posted Q4 net revenue of $333.1M (+2% YoY), with international revenue surging 65% to $50.7M and full-year 2025 reaching $1,268.1M. Adjusted EBITDA hit $274.7M (21.7%); Florida expanded to 70 dispensaries and the QMID device launched in the UK and Germany.
- The company completed a US$500M senior notes offering at 11.5% — the largest in US cannabis history — refinancing $457M due December 2026 with maturity extended to February 2029. Curaleaf also announced proposed domestication to Delaware.
- Green Thumb Industries reported Q4 revenue of US$311.1M (+5.7%), FY2025 of $1.18 billion (+3.4%) and GAAP net income of $114.2M across 113 RISE dispensaries in 14 states. Minnesota‘s adult-use launch drove growth, though gross margin declined to 48.9%.
- GTI secured an additional $50M in senior debt, bringing the total facility to $189M (September 2029 maturity).
- Trulieve posted FY2025 revenue of $1.18 billion (flat YoY) with a 60% gross margin, record adjusted EBITDA of $427M (36%) and record free cash flow of $229M. The company redeemed $368M in notes, holds 233 dispensaries and received a conditional Texas licence.
- Verano launched the Swift Lifts pre-roll brand across five states, tapping a segment that grew 22% in 2025. The company upsized its revolving credit facility from $75M to $100M (February 2029 maturity, SOFR+6%) and opened MÜV Deltona — its 83rd Florida dispensary and 160th nationwide.
- GoldFlower Cannabis received approval for a new cultivation site in Florida and partnered with Heavyweight Heads genetics.
- Jazz Pharmaceuticals posted record $4.3 billion total revenue; Epidiolex crossed $1.1 billion in annual sales (+9%) while Sativex declined to $16.3M.
- MedMen‘s court-appointed receiver filed a $1B+ damages suit against Serruya, Superhero and Tilray, arguing the company’s peak $3 billion capitalisation and the “option value” of federal reform support the claim.
- Chicago Atlantic closed financing for S1 Enterprises, the parent of Illicit Cannabis, enabling a 100% ESOP for 500+ employees across Missouri and New Jersey — a tax-exempt S-Corp structure rare in cannabis.
- Cannabist Company closed a $130M sale of Virginia assets to a Millstreet affiliate, comprising five dispensaries and 82,000 sq ft of cultivation. The deal replaced a prior $110M agreement with Curaleaf.
- Nature’s Miracle signed a $150M MOU for a California facility covering 660,000 sq ft with 40 MW solar, targeting production costs below $200/lb.
- Planet 13 completed its exit from California, divesting retail, distribution and cultivation to focus on Nevada and Florida.
- iAnthus extended US$8.4M in bridge notes to June 2027, plans a 26th Florida dispensary in Tequesta and confirmed The Vault brand expansion into New Jersey.
State-level data reinforced the bifurcation. Massachusetts reached a $10 billion cumulative sales milestone. Nevada reported FY25 taxable sales fell 8.6% to $758M, with ~$96M transferred to the state education fund. Michigan‘s recreational sales dropped 16% in January as the new 24% wholesale tax, harsh winter and a December pre-buy surge combined to depress demand.
- BDSA estimated January sales fell 3.1% sequentially to $2.07 billion across 15 states, though year-on-year growth held at +4.6%.
- The CannabisDeals US Cannabis Price Index showed modest retail inflation to 101.9; pre-roll prices plummeted 62% to $17.11, THC flower held at $62.35, and CBD faced structural 15–30% discount pressure.
- New York‘s cannabis market continued to expand, closing 2025 with its first half-billion-dollar quarter. Illinois reported December adult-use sales of $116.6M (+5.6% MoM), though full-year 2025 fell 12.5% to $1.51 billion — below 2022 levels.
- A Higher Origins report found California cultivation in broad decline: outdoor peaked in 2022, Santa Barbara and Humboldt account for 44% of state canopy, and indoor showed its first cracks in 2025.
The hemp-derived THC beverage segment surged. Willie’s Remedy+ raised a $15M Series A led by Left Lane Capital, having sold 400,000+ bottles in under a year and claiming #1 THC beverage online. A 10-SKU portfolio is rolling out through Total Wine, Lowe’s and Binny’s via JuneShine Brands‘ three-tier wholesale network.
- An analysis argued US alcohol faces structural compression, not THC disruption — current THC revenue growth primarily reflects black-market migration into legal reporting rather than displacement of drinking occasions.
- Ska Brewing launched Lo Dose tonics in Colorado (1.5 mg THC / 25 mg CBD), while Saucy Seltzer partnered with Blue Ridge Spirits for national distribution from its Ohio facility with up to 2.75M cases of annual capacity.
- Delta Beverages added a cannabis spirit (10 mg THC / 1.5 oz serving, $59/bottle).
- Azuca marked eight years and 700M dosed servings via its TiME INFUSION platform across 30 US states, Puerto Rico, Canada and Australia.
- Ska Brewing launched Lo Dose tonics in Colorado (1.5 mg THC / 25 mg CBD), while Saucy Seltzer partnered with Blue Ridge Spirits for national distribution from its Ohio facility with up to 2.75M cases of annual capacity.
IP and infrastructure signals pointed to sector maturation. Natural Extraction Systems filed patent infringement suits against multiple THC manufacturers over four distillation processes. Safe Harbor expanded cannabis payment solutions, and True Terpenes launched Headstash, a compliant cannabis aroma product (<0.3% THC. A growing exodus of cannabis executives underscored shifting workforce dynamics.
- Cannabis stocks continued to decline amid volatility, regulatory uncertainty and investor reassessment of sector exposure. Cannabis Business Times reviewed 2025 lessons and 2026 outlook across market trends, regulatory developments and growth opportunities.
🔬 Science — Kaiser Permanente Cohort Links Adolescent Cannabis To Psychosis Risk
A Kaiser Permanente Northern California cohort of 463,396 adolescents found that cannabis use before age 18 was associated with significantly higher rates of psychotic and bipolar disorder diagnoses in young adulthood — the largest epidemiological signal linking adolescent cannabis exposure to serious psychiatric risk this cycle.
- Anxiety, mood and sleep — A 30-day ecological diary (n=345) found CBD-dominant products yielded larger anxiety reductions than THC-dominant ones, while a 45-day Releaf/CannaMD Florida cohort (n=416) reported the largest anxiety relief from medical cannabis among outcome measures.
- A crossover pilot explored cannabinoid supplementation for subthreshold insomnia and mood disturbance.
- An Oregon Health & Science University trial (n=18) tested THC administered before bedtime on sleep architecture.
- Pain and pharmacology — A PNAS study identified Nav1.8 as an analgesic target for nonpsychotomimetic cannabinoids, offering a pathway to pain relief without psychoactive effects.
- An RCT (n=54) found no benefit from oral CBD for TMJ pain.
- Select terpenes demonstrated antinociceptive activity via adenosine A2a receptors.
- A Healio orthopaedic survey found 81% of patients amenable to THC and 90% to CBD for musculoskeletal pain.
- Novel therapeutics — Charlotte’s Web‘s DeFloria subsidiary received FDA clearance for a Phase 2 ASD trial of botanical AJA001.
- The NIH-funded LiBBY Study at the University of Kentucky is investigating cannabis for late-stage dementia agitation.
- Silvanex, combining silver with CBC and CBG, showed promise against drug-resistant infections.
- An RCT (n=120) found cannabis broadly impairs multiple memory domains, adding nuance to cognitive safety assessments.
- Dosing standards — The Index of Cannabis Equivalence (ICE) established standardised dose equivalencies across administration routes (n=1,368): 2 puffs ≈ 1 bong hit ≈ 5 mg THC edible (Journal of Psychoactive Drugs).
- Psychiatric risk — A review found THC linked to negative mental health outcomes while CBD showed psychiatric therapeutic potential. The UT Dallas Filbey lab (n=395) mapped cannabis and depression effects on brain topology.
- A JAMA Network Open study examined brain function outcomes of cannabis use in young adults.
- Among veterans, combined cannabis and tobacco use was associated with suicide attempt and overdose death risk.
- Substance use patterns — Tobacco Reporter documented rising cannabis–nicotine dual use.
- A cross-sectional study (n=549) examined cannabis and erectile dysfunction in sexual minority men.
- A JAMA Psychiatry cross-sectional study compared medical versus nonmedical cannabis use among US adults.
- Pregnancy and youth — US teen survey data showed adolescent drug use declined across most substances except nicotine pouches, which doubled in 2024.
- A claims analysis (2015–2020) quantified cannabis use disorder among insured pregnant women.
- PRAMS data (n=802,954) explored motivations for cannabis use during pregnancy.
- Crime and utilisation — A difference-in-differences analysis using FBI UCR data examined marijuana legalisation and crime across 50 states.
- Among Northern California cancer patients (n=2,602), 24.7% reported using cannabis.
- Respiratory research found combustion elevates airway metabolites versus e-devices in vaping and marijuana users.
- A historical study of Bengal (1911–1925) analysed substitution between alcohol, cannabis and opium.
- Collins (2026) assessed clinical risks and regulatory gaps in safeguarding cannabis for medical use.
- A prospective mixed-methods study examined the COVID-19 pandemic’s effect on cannabis demand.
- Research policy and funding — The Trump administration’s proposed cuts threaten 565 active NIH cannabis experiments worth over $310M and raise rescheduling questions.
- The University at Buffalo partnered with Pure Jamaican and DENT Neurologic Institute for multinational cannabinoid R&D.
- California’s DCC awarded ~$30M in cannabis tax-funded research grants covering THC drinks, terpenes and tribal cannabis sales.
- David Watson (“Sam the Skunkman”), legendary cannabis breeder, died — a foundational figure in modern cannabis genetics.
- HashDash survey (n=500+) — Stress and anxiety relief drive cannabis use: 71% consume for stress, 54.8% for anxiety, yet 60% fear cannabis may worsen anxiety.
- Breeding — A study of hemp cultivars (>1,300 measurements) revealed heritable high-CBC sub-chemotypes with significant breeding potential.
- Analysis of 909 feral cannabis samples identified adaptive variation for climate-resilient breeding.
- Postharvest trials showed drying and curing methods affect cannabinoid content in industrial hemp.
- Industrial hemp and sustainability — A Georgia Tech cost-energy-emissions analysis found hemp insulation could make buildings greener and create US construction jobs.
- Complementary research explored wood–hemp–sodium silicate composites for additive manufacturing and waste hemp hurd reinforced PRF composites for construction.
- A One Earth study documented indoor cultivation’s expanding carbon footprint as the industry scales.
🇦🇺 Australia — LGP–Cannatrek Merger Creates A$112M Champion As TGA Reform Accelerates
🔭On the radar: TGA discussion paper submission to ACMS in March 2026; MCIA/AMCA second public consultation on SAS reform in April 2026.
🏛️ Regulation — 790 Submissions Find SAS Framework “Not Fit-For-Purpose” As RACGP Demands ARTG Registration
Australia’s TGA will submit a discussion paper to the ACMS in March after 790 submissions on unapproved medicinal cannabis found broad agreement that the current SAS/AP framework is “not fit-for-purpose.” The RACGP called for mandatory ARTG registration within 18–24 months, citing major public health risks from THC-dominant products prescribed predominantly to the 18–44 age group.
- The RACGP submission urged PIC/S GMP, clinical trials, and a crackdown on telehealth-only prescribing services that “circumvent TGA regulations.” Only two medicinal cannabis products are currently ARTG-approved despite hundreds of thousands of patients accessing unapproved cannabis via SAS/AP.
- Novachem proposed a dedicated ARCTG register with AUST C identifiers, mandatory batch testing, a 50 mg/day THC guidance cap for Schedule 8, and transition from SAS/AP to registered supply by January 2027.
- MCIA and AMCA launched an “evolution not revolution” campaign urging the TGA to preserve patient access, citing a 0.06% adverse event rate. Stakeholder workshops began in February; a second public consultation is planned for April 2026.
- Cannabis Clinicians Australia will join the ANZCCP through a planned merger, departing from AMCA — restructuring the practitioner body landscape ahead of reform.
- Tasmania implemented interstate prescribing rules for controlled medicines effective 16 February 2026, allowing pharmacists to dispense interstate cannabis prescriptions and removing the prohibition on bringing medicines into the state.
- Victoria‘s $5.2M closed-track medicinal cannabis driving trial delayed to 2027 and will no longer assess impairment testing technologies, undermining the Swinburne University programme’s original scope.
- The ODC issued a $19,800 fine to Hale Farm for failing to follow waste-disposal SOPs under its medicinal cannabis licence — signalling tighter enforcement under the Narcotic Drugs Act.
💼 Market — Pastilles Reach 30% Of SAS-B Prescriptions As Canadian Entrants Expand Supply
International brands and non-flower formats reshaped Australia’s supply in February. SAS-B data confirmed pastilles at almost 3 in 10 approved prescriptions in January — accelerating the format’s displacement of oils as prescribers favour dosing precision.
- M&A and corporate restructuring dominated the cycle. The LGP–Cannatrek merger progressed toward scheme meetings on 2 April with implementation targeted 1 May; the combined group’s pro-forma revenue stands at A$112m.
- LGP divested its WA facility for A$7.8m in a sale-and-lease-back to fund expansion. ⏳ Scheme meetings 2 April; implementation 1 May.
- Cannim entered formal liquidation after creditors voted to wind up the group. Olvera Advisers identified A$32m in potentially recoverable transactions including A$9m in uncommercial related-party advances and a Jamaican property sold at US$1.5m against a US$6.3m listing.
- Liquidator set 1–3 month timeframe for director examinations and demanded return of A$465,644 in preferential payments; insolvency indicators from August 2024. Three UK subsidiaries remain outside the process. ⏳ Public examinations 1–3 months; legal proceedings 4–8 months.
- Argent BioPharma reported 98% collapse in customer receipts (A$133k → A$2k) in Q4 2025, surviving via A$3.1m equity raise.
- AusCann acquisition stripped from US$15m to CannPal stake and Neuvis option for 20m shares at A$0.10.
- Canadian entrants expanded supply: Organigram launched 10 Edison and BOXHOT vapes and pastilles to 4,000+ pharmacies via Leafio.
- Nextleaf Solutions shipped 756,000 softgels to a white-label client in January.
- Aurora Cannabis launched new THC flower and resin cartridges in Australia and New Zealand.
- Earnings bifurcated between scaled manufacturers and margin-pressured distributors.
- Bioxyne posted record H1 FY26 revenue of A$31.3m (+149%) with A$8.3m adjusted EBITDA (+137%), upgrading full-year guidance to A$16.5–19.0m. Four new GMP clean rooms lifted flower processing capacity 150%, Australian customers grew 104%.
- Also received first GMP psilocybin capsules order — 250 doses for ~60 TRD patients in Queensland and WA — extending into psychedelic therapeutics via Breathe Life Sciences.
- Vitura Health grew revenue 8% to A$67.9m but swung to net loss of A$0.8m as price compression pushed gross margins to 22.7%.
- Elixinol reported FY25 revenue of A$15.5m (+3.6%) with gross margins expanding to 39% and first underlying operating cash-flow-positive quarter.
- Epsilon Healthcare posted record December-quarter receipts after ASX reinstatement.
- Neurotech International lifted cash to ~A$6.3m after A$4.73m R&D tax refund and secured Phase 3 ethics approval for NTI164 in ASD.
- Bioxyne posted record H1 FY26 revenue of A$31.3m (+149%) with A$8.3m adjusted EBITDA (+137%), upgrading full-year guidance to A$16.5–19.0m. Four new GMP clean rooms lifted flower processing capacity 150%, Australian customers grew 104%.
🔬 Science — CBN Insomnia RCT Misses Primary Endpoint But 300 mg Signals Dose-Dependent Potential
The first RCT of cannabinol for diagnosed insomnia at the Woolcock Institute of Medical Research found the primary endpoint was not met — neither 30 mg nor 300 mg CBN significantly reduced wake after sleep onset — but 300 mg demonstrated significant improvements in subjective sleep quality (p=0.005), reduced sleep onset latency (p=0.004) and increased NREM-2 sleep (p=0.03) across 20 adults.
- Pain therapeutics advanced on multiple fronts: Nexalis Therapeutics (formerly InhaleRx, ASX: NX1) is preparing a pivotal trial for breakthrough cancer pain, targeting a therapeutic void in end-of-life analgesia.
- Nexalis outlined its 2026 clinical plan in its first investor presentation under the new ticker NX1; shares climbed 35% to 3.0 cents.
- A case report documented sustained phantom limb pain relief in a 64-year-old amputee using liposomal β-caryophyllene as adjunct to high-THC cannabis — pain dropped from 9/10 to ≤6/10 over two years with no tolerance.
- A methodological review examined design considerations for cannabis trials in palliative care settings.
- Epidemiological and practice evidence reinforced the safety signal as TGA reform accelerates: NSW Poisons Information Centre data (2014–24) showed cannabis poisonings increased but found no trend change at the 2015–16 medicinal rescheduling.
- An ACT study mapped home-grown cannabis cultivation behaviours post-decriminalisation, profiling consumer patterns under Australia’s only personal cultivation framework.
- A review analysed the evolution of medicinal cannabis policy and regulatory oversight, drawing lessons for sustainable integration.
- A national survey of 730 pharmacists found only 5.5% initiated deprescribing discussions for medicinal cannabis — far below opioids (48.2%) and benzodiazepines (42.5%) — with 84% requesting further training.
🇨🇦 Canada — Organigram Posts 46% Revenue Growth As Auxly/Ayurcann CCAA Signals Consolidation Wave
🔭On the radar: Nova Scotia Bill 200 cannabis enforcement second reading pending; Alberta 2% early payment fee elimination on 1 April 2026.
🏛️ Regulation — Health Canada Publishes CBD NHP Report As RCMP Flags Organised Crime
Health Canada published its “What We Heard” report on health products containing CBD, summarising stakeholder feedback after removing the CBD Natural Health Product regulation from its forward plan. The report provides the first consolidated view of industry and patient positions on a CBD product pathway, but with the NHP proposal shelved, the regulatory route for CBD products in Canada remains undefined.
- NORML Canada formally criticised petition e-6991 — sponsored by Conservative MP Dean Allison — which proposes raising the cannabis access age to 25, banning edibles and revoking producer licences over odour. Executive director Jennawae Cavion argued the proposals lack evidence and would expand illicit markets; 800+ signatures as of 11 February. ⏳ Open until 9 April 2026.
- A second petition (e-6874), sponsored by NDP leader Don Davies, called for a Pan-Canadian Cannabis Framework with intergovernmental oversight and harmonised standards; 167 signatures, closed 26 February 2026.
- The RCMP published “The blunt truth” on 17 February, detailing how criminal organisations exploit Canada’s cannabis market — citing a 2024 joint New Brunswick–Québec seizure of 432 kg bound for 30+ unlicensed dispensaries and a January 2026 CBSA interception of 1,400 kg for illegal export.
- Nova Scotia Attorney General Scott Armstrong introduced Bill 200 on 25 February to expand enforcement authority, increase fines and penalise landlords permitting illegal cannabis activity — targeting over 100 illegal outlets against just 51 legal NSLC stores. ⏳ Second reading pending.
- Health Canada refused or revoked over 4,100 registrations under its PPL/DG programme as of March 2025, including ~3,400 for public health reasons — up from 1,400 in late 2024. The regulator conducted 889 inspections in 2024–25 and revoked 90 commercial licences in 2025.
- Alberta will eliminate the 2% early payment fee for producers effective 1 April 2026, announced alongside Budget 2026; cannabis tax revenue projected at $229M in 2026–27.
- StratCann examined temporary foreign worker practices in the cannabis sector, raising scrutiny over labour conditions as the industry scales production.
- A Health Canada–commissioned study found 48% of licensed products deviate more than 20% from labelled THC content, while 94% of illicit samples contained pesticides — reinforcing the regulated quality gap. Seven lots totalling 2,916 units from Great White North Growers were recalled for arsenic.
- Health Canada issued updated sampling guidance for federal licence holders, mandating storage at licensed sites, traceability documentation and compliance enforcement; provinces retain final authority on retailer sample acceptance.
💼 Market — C$5.62bn Record Retail Year As Organigram Surges 46% And Two LP Groups Enter CCAA
Canadian LPs posted broad-based revenue growth in February — Organigram surging 46%, Cronos hitting a record quarter (+47%) and High Tide crossing C$164M (+19%) — as the retail market reached a record C$5.62 billion in calendar 2025 (+4.1%). Two fresh CCAA filings and Canopy Growth‘s court-approved acquisition of MTL Cannabis confirmed that consolidation is now outpacing topline expansion.
- Canadian cannabis retail sales hit a record C$503.7M in December 2025 (+2.9% YoY), bringing full-year 2025 to C$5.62 billion (+4.1%). Statistics Canada launched the Industrial Product Price Index for cannabis at 135.7 (January 2020 = 100).
- A market share analysis showed no company above 25%, with Organigram leading and average EBITDA-to-sales margins at 20–25%.
- A StratCann investigation found Canadian LP exports surging as domestic saturation and excise taxes exceeding 37–50% push revenue offshore: Aurora posted $48M in international sales, Pure Sunfarms grew +772% and Decibel +2,621%.
- The industry sought government support for exports, arguing federal trade infrastructure lags commercial readiness. Tilray signed a five-year US beer licensing deal with Carlsberg Group, starting January 2027 — a major diversification beyond cannabis.
- Canopy Growth‘s court-approved acquisition of MTL Cannabis and two fresh CCAA filings confirmed that consolidation is outpacing topline expansion, even as the largest LPs posted record or near-record quarters.
- Canopy Growth reported Q3 net revenue of $75M (flat YoY): cannabis $52M (+4%) and medical $23M (+15%), offset by international declining 31% to $6.2M. Net loss narrowed 49%; a $185M recapitalisation extended maturities to 2031.
- MTL Cannabis shareholders voted 99.97% in favour of the Canopy Growth acquisition (89% participation), with final court approval on 23 February. Closing is expected before end of March 2026. ⏳ Pending.
- Ayurcann (parent of Auxly) filed for CCAA creditor protection on 30 January, disclosing CRA tax arrears of C$10.6M. Auxly established a $4.64M stalking horse bid on 20 February; bids are due 31 March. ⏳ Pending.
- Simply Solventless — parent of MHF, CannMart and ANC — commenced restructuring under CCAA with MNP Ltd as monitor, the second LP group to enter creditor protection in the cycle.
- Avant Brands adopted a shareholder rights plan, signalling defensive positioning amid rising M&A activity.
- Canopy Growth reported Q3 net revenue of $75M (flat YoY): cannabis $52M (+4%) and medical $23M (+15%), offset by international declining 31% to $6.2M. Net loss narrowed 49%; a $185M recapitalisation extended maturities to 2031.
- Organigram reported Q1 FY2026 gross revenue of C$97.3M (+46% YoY) with net revenue of C$63.5M (+49%) and adjusted EBITDA of C$5.3M (+273%). International revenue climbed 51% to C$5.0M under new CEO James Yamanaka.
- A Forbes analysis found M&A shifting to private deals — consistent with the sector’s retreat from public-market transactions. Sensi Brands CEO Tony Giorgi discussed global expansion strategy.
- Tilray posted record Q2 net revenue of $218M, with cannabis up 3% YoY and net loss narrowing 49%.
- Cronos delivered Q4 record net revenue of $44.5M (+47%), lifting full-year 2025 to $146.6M (+25%) with adjusted EBITDA positive at $10.1M. The company holds $832M cash; the CanAdelaar acquisition remains pending. ⏳ Pending.
- High Tide posted Q4 record revenue of $164M (+19%) with adjusted EBITDA of $12.4M (+51%) across 218 Canna Cabana locations and 2.5 million club members. The company entered Germany through Remexian.
- Nextleaf Solutions reported FY2025 gross revenue of $14.96M with positive EBITDA of $522K and net loss cut 88.6% — a turnaround signal for the extraction specialist.
- Mercanto Holdings posted Q1 revenue of C$900K (+9% YoY), expanding vape categories in Québec with no long-term debt.
- Cannara Biotech graduated from TSXV to TSX as TD Cowen initiated coverage; the company operates 1.6 million sq ft with 100,000 kg annual capacity.
- Decibel Cannabis closed a $61M credit facility with ATB Financial at a five-percentage-point interest rate cut.
- Decibel sold its Creston property for C$2.5M and consolidated cultivation at Battleford — projecting C$4M in annual savings.
- Village Farms upsized its cannabis credit facility by C$15M, extending maturities to February 2029.
- Avicanna closed a non-brokered private placement. Aurora Cannabis filed a US$100M ATM share offering.
- MediPharm Labs announced a CEO transition: David Pidduck stepped down with CFO Greg Hunter as interim.
- Cronos launched Spinach PUFFERZ, a palm-style all-in-one vape using liquid diamonds at 98.1% THC in three flavours — available in Alberta, British Columbia and Ontario, with national rollout in April 2026.
- PAX re-entered the Canadian market with PAX TRIP live rosin all-in-one devices in Ontario, Manitoba and Saskatchewan via CannaPiece Corp.
- Rubicon Organics completed the first harvest at Cascadia in Hope, British Columbia — adding 4,500 kg annually (+40%) from the 47,500 sq ft expansion, with monetisation expected in Q2 2026.
🔬 Science — CAMH Ranks Alcohol Most Harmful; Cannabis Scores 15 Of 100
A CAMH-led panel of 20 experts scored 16 substances across 16 harm dimensions, ranking alcohol as Canada’s most harmful drug at 79/100 — ahead of tobacco (45), non-prescription opioids (33) and methamphetamine (19). Cannabis scored 15, reinforcing the disparity between alcohol’s population-level harm and its regulatory treatment.
- Psychiatric and public-health risk intensified alongside legalisation. A JAMA Network Open cohort of 13M+ Canadians aged 14–65 examined changes in CUD-associated schizophrenia risk after medical and recreational legalisation, while an Ontario study found hospital-based CUD patients at markedly increased 5-year mortality.
- An Ontario natural experiment used the 2019 retailer licence lottery design to assess recreational retailer density’s impact on emergency department visits.
- A sustainability analysis modelled cannabis cultivation health externalities, estimating cannabis-associated mortality and exploring food-crop reallocation scenarios.
- Clinical outcomes reinforced therapeutic signals but fell short of clinical thresholds. A 24-week MCRWE study of 276 patients via Shoppers Drug Mart reported significant reductions in pain interference (PROMIS −4.6), anxiety (GAD-7 −2.24) and depression (PHQ-9 −2.79), though no outcome met its minimal clinically important difference.
- Avicanna published real-world evidence results supporting its medical cannabis product efficacy.
- THCV/CBD mucoadhesive strips showed statistically significant weight loss, decreased systolic blood pressure and LDL cholesterol — an emerging metabolic therapeutic pathway.
- Preclinical evidence showed CBD potentiates HPA axis responsivity with sex-specific effects in mice, advancing the mechanistic case for cannabinoid anxiolysis.
- Quality and safety — Health Canada compared 50 licensed and 50 illicit samples, finding 48% of legal products deviated >20% from labelled THC while 94% of illicit samples contained pesticides averaging 3.4 compounds per sample.
- A separate risk assessment found low health risk from heavy metals in regulated cannabis, with consumption well below permissible daily exposures.
- The most extensive multi-omic analysis of ethylene-induced sex change in Cannabis sativa integrated 130+ RNA-seq libraries across three genotypes, identifying 14 high-confidence candidate genes — a step toward sex-stable cultivars for commercial seedless production.
🇧🇷 Brazil — Anvisa Publishes RDC 1,015: Domestic Cultivation Authorised For 4M-Patient, R$1bn Market
🔭On the radar: RDC 1,015 takes effect 4 May 2026.
🏛️ Regulation — Anvisa’s RDC 1,015 Authorises Domestic Cultivation For 4M-Patient Market
Anvisa published RDC No. 1,015/2026 on 3 February — Brazil’s most comprehensive medical cannabis overhaul — restricting authorisation to CBD-based pharmaceuticals (0.2% THC limit), imposing five-year transitional sanitary authorisation with mandatory drug registration at expiry, and tightening labelling, traceability and pharmacovigilance. Separately, the agency unanimously approved domestic hemp cultivation rules (≤0.3% THC), targeting 4 million patients in a R$1 billion market, after the STF ruled it incoherent to permit imports while prohibiting national production. ⏳ RDC 1,015 effective 4 May 2026.
- RDC 1,015 excludes cosmetics, food and non-pharmaceutical categories, bans terms such as “CBD oil,” “full spectrum” and “broad spectrum,” and requires warnings that efficacy has not been evaluated by Anvisa. Prescribing is restricted to physicians and dentists with mandatory informed consent.
- The resolution reclassifies CBD from black-label to red-label (schedule A1 → C1), enabling digital prescriptions via the SNCR system for products with up to 0.3% THC. Teleconsultations and remote dispensing through major chains — Raia, Drogasil, Panvel — become possible for the first time.
- Brazil’s importation crossroads intensifies as ~900,000 patients already use cannabis extracts and pharmacy prices reach R$2,000 for a 30 ml bottle — domestic production is expected to drive significant cost reductions.
- The cultivation framework drew on a 2023 Anvisa delegation to Colombia and a prior regulatory sandbox for artisanal cosmetics. Director Thiago Campos described a programmatic norm — general guidelines with technical detail deferred to a working group — that achieved unanimous board approval after 30 days of intensive negotiation.
- The government, encouraged by positive reception, aims to advance industrial hemp regulation by removing hemp from the Portaria 344 prohibited substances list before the electoral calendar closes. ⏳ Hemp delisting targeted February–June 2026.
- Implementation concerns surfaced around the 0.3% THC cultivation limit: researchers noted only ~10% of Cannabis sativa varieties fall below this threshold, and Brazil’s tropical climate — with high year-round solar exposure — tends to boost cannabinoid production beyond temperate-origin seed specifications.
💼 Market — ALKO Do Brasil Builds Medicinal Cannabis Unit As QuickStrip Enters With Oral Film
Brazil’s commercial cannabis infrastructure expanded as pharmaceutical companies moved to capitalise on the RDC 1,015 framework. ALKO do Brasil launched a dedicated medicinal cannabis unit with structured specialist leadership — appointing Leontino Vieira Junior as commercial head and Dennys Zsolt Santos as Head of Operations — starting with CBD products and planning gradual portfolio expansion aligned with regulatory evolution.
- Canadian biotech Rapid Dose Therapeutics commercially launched QuickStrip oral thin film in Brazil through partner Entourage Phytolab, becoming among the first to introduce dissolvable strip cannabis therapy to the market following Anvisa’s approval of sublingual and buccal routes. The company cited 672,000+ registered patients (+56% YoY), ~50,000 prescribing doctors and a market projected to reach US$1.5 billion by 2034.
🔬 Science — Meta-Analysis Of 21 RCTs Confirms CBD Efficacy For Anxiety Disorders
A meta-analysis of 21 placebo-controlled RCTs found pure CBD produced a moderate anxiolytic effect (SMD −0.61) and THC-enriched formulations a comparable signal (SMD −0.65), though overall pooled cannabinoid results were non-significant (SMD −0.40). Evidence quality remains low; authors call for larger double-blind trials before routine clinical use.
- Preclinical and analytical research advanced on two fronts: a UFU study evaluated antimicrobial activity of cannabidiol, demonstrating CBD’s potential as a therapeutic alternative against bacterial infections.
- A novel electrochemical method for detecting synthetic cannabinoids in e-cigarette and biological samples was developed using boron-doped diamond electrodes — the first comprehensive voltammetric investigation of AB-CHMINACA.
- Research infrastructure expanded as GreenCare Pharma became the first Brazilian pharmaceutical company authorised to research cannabis extract-based medications, while competitors remain focused on isolated CBD.
- Abrace (50,000+ members) and the Federal University of Rio Grande do Norte opened dialogue on research partnerships targeting analytical quality control and new formulations under Anvisa‘s revised regulatory framework.
🇬🇧 United Kingdom — FSA Publishes First CBD Consumer Survey As Record 137t Seizure Exposes Illicit Scale
🔭On the radar: FSA novel food full authorisations expected by autumn 2026.
🏛️ Regulation — Drug Science Challenges ACMD Scheduling As Crown Court Clarifies Section 5A Defence
The FSA published its first CBD consumer behaviour survey, revealing high usage and reliance but poor regulatory awareness — full novel food authorisation is expected by autumn 2026. A record 137 tonnes of herbal cannabis seized in 2024/25 exposed the illicit market’s persistent scale, while Drug Science challenged the ACMD‘s cannabis scheduling position.
- Novel food governance came under scrutiny as Hemp Hound filed an Article 4 challenge against the FSA over its filing process, with 11,456 CBD products on the register amid concerns over transparency and approval consistency.
- The FSA was described as “finding its stride” on novel food oversight, though the sector awaits clarity on enforcement timing and product approvals.
- Drug Science formally challenged the ACMD’s scheduling position on cannabis-based products for medicinal use, arguing the evidence base has evolved since the 2018 rescheduling.
- The College of Policing published APCDLO guidance confirming medical cannabis as a Schedule 2 medicine, but no dedicated officer training is planned despite a coroner’s finding highlighting knowledge gaps. The Patient Protect service launched to help patients verify legal status.
- Sal Aziz was fully acquitted at Winchester Crown Court (10 February 2026) of drug-driving charges after a two-year legal battle. The judge dismissed claims that medicine used past 30 days or its expiry date was unlawful, clarifying the Section 5A statutory medical defence for the UK’s 90,000+ medical cannabis patients.
- Home Office data showed 269,321 drug seizures in the year to March 2025 — the highest since records began in 1973. Cannabis accounted for 194,682 seizures (+28% YoY), with Border Force contributing 127 of the 137 tonnes (+71%) via 65 individual seizures exceeding 100 kg.
- Some 430,000 cannabis plants were seized; approximately 76% of plant seizures involved 10 plants or fewer.
💼 Market — Tilray Targets £1bn UK Pharma Category As International Operators Accelerate Entry
International operators accelerated UK market entry in February. Tilray‘s distribution arm CC Pharma signed a strategic agreement with Smartway Pharmaceuticals to expand parallel import and specialist pharmaceutical supply across a category estimated at nearly £1 billion — a deal designed to embed Tilray’s own medicinal cannabis products in the UK health system.
- Curaleaf Laboratories and Sanity Group partnered to develop and manufacture medical cannabis products for UK patients, combining Curaleaf’s MHRA-approved infrastructure with Sanity Group’s pharmaceutical IP. First products expected on prescription from Q1 2026.
- California-based Sunderstorm partnered with Grow Group UK and New Garden Pharma to bring the Kanha edibles brand to UK patients via EU-GMP manufacturing in North Macedonia — including the first premium Thai-grown flower in the UK market.
- VOR Pharma launched Foundation Medical, a three-formulation portfolio via specialist prescription and the MYPAS scheme. Auravia Medical introduced the UK’s first cannabis clinic for NHS waiting list patients, with reduced £30 monthly fees and a relaunched Grow Access Project discount scheme.
- German operator enua secured UK licences and expanded local operations, positioning the UK — Europe’s second-largest medical cannabis market — as a long-term strategic commitment built on local infrastructure.
- Breathe Life Sciences (Bioxyne subsidiary), which built Australia’s pastilles category from <5% to >25% of SAS-B prescriptions in 18 months, secured a Scottish Borders manufacturing site with £848k SOSE funding — targeting 3 million pastilles/month and up to 100 jobs within three years. MHRA resistance to pastilles as a dose form remains the key regulatory barrier.
- Bloem Teknik secured £700k+ Innovate UK funding for an autonomous greenhouse lighting R&D project applicable to controlled-environment cannabis cultivation.
- Cannaray Brands was named Official Wellness Partner for Spartan Race UK and Tough Mudder UK in a three-year deal (2026–2028), extending cannabis-adjacent branding into mainstream fitness events.
- Plantz.io launched Cannavec.ai at Cannabis Europa Paris — a commercial API providing clinics and platforms access to the Cannabis Knowledge Foundation‘s evidence-based cannabis knowledge base.
🔬 Science — Researchers Propose ‘THC Units’ As First Weekly CUD-Risk Thresholds
University of Bath researchers used the CannTeen longitudinal study (n=150) to propose the first weekly THC-unit thresholds for cannabis use disorder risk: ~8 units/week for adults and ~6 for adolescents (1 unit = 5 mg THC), with 86–91% sensitivity. Near-identical adolescent cut-offs for any versus moderate/severe CUD suggest no safe dose–response gradient for younger users.
- Clinical evidence advanced on two fronts. A two-year case series from the UK Medical Cannabis Registry assessed clinical outcomes in depression, contributing real-world longitudinal evidence as the UK prescribing base approaches 100,000 patients.
- Preclinical research found low-dose THC combined with a common anti-inflammatory holds promise in Alzheimer’s treatment — an emerging therapeutic pathway beyond established pain and anxiety indications.
- Policy and medical education progressed in parallel. A comparative analysis found legal cannabis regulations protect public health more effectively than alcohol laws, reinforcing the regulatory-disparity argument as the UK debates scheduling reform.
- Bristol Medical School approved the first medical cannabis teaching placement for Year 3 MBChB students — proposed by Dr David Tang via the MCCS — covering prescribing frameworks, clinical indications and the endocannabinoid system.
- 🇮🇲 Public Health Isle of Man commissioned Liverpool John Moores University to conduct an island-wide survey on medicinal cannabis use, alcohol consumption and illicit drug patterns — the first systematic data collection since legislation permitted prescribed cannabis.
- The survey (open 4 February – 18 March 2026) will be benchmarked against the 2022 Substance Use Survey, which found 25.8% of adults had used at least one substance in the prior 12 months; Director of Public Health Dr Matt Tyrer said findings will feed into a report later in 2026. ⏳ Survey closes 18 March; report expected H2 2026.
🇳🇱 Netherlands — All 10 Wietexperiment Growers Now Active But No Budget For Post-Evaluation Legislation
🔭On the radar: ChristenUnie/SGP amendment vote to phase out Wietexperiment — repeatedly postponed, still pending.
🏛️ Regulation — 68-Seat Legalisation Majority Secures Experiment As Coffeeshop Closures Erode Footprint
The new D66–VVD–CDA minority coalition committed to continuing the Wietexperiment but allocated no budget for post-evaluation legislation. 68 Tweede Kamer seats explicitly back legalisation — 8 short of a majority — securing the experiment politically while leaving structural supply-chain legislation unresolved before the August 2028 evaluation deadline.
- The ChristenUnie, SGP and Groep Markuszower (13 seats) filed an amendment to the Justice and Security budget to phase out the experiment, citing LADIS data showing cannabis accounts for 39% of under-25 addiction-care cases. The vote was repeatedly postponed; passage is unlikely given the pro-legalisation majority. ⏳ Vote pending.
- The Bond van Cannabis Detaillisten challenged Worldline’s collective termination of coffeeshop payment contracts — initiated 20 November 2025 without individual compliance failures — arguing it violates anti-money-laundering principles and Dutch Finance Ministry guidance banning categorical exclusion.
- Local implementation diverged: VVD Tilburg proposed a cannabis drive-through at the city’s edge in its March 2026 election manifesto, while Groningen lost two more experiment coffeeshops.
- De Dees and Driemaster will permanently close on 1 May 2026 after owner Marja Plas lost her appeal against a Bibob-based licence revocation — reducing the city from 14 (1992 cap) to 7 coffeeshops and extending a string of closures shrinking the experiment’s operational base.
- IMC Nederland urged the incoming health minister to improve medical cannabis access after two Trimbos Institute studies documented barriers including limited prescribers, high costs and stigma — 500,000 adults self-medicate versus only 7,000 on prescription.
- Bedrocan opened direct industrial orders for parties with valid opium exemptions, the first step in the medical framework restructure as the BMC‘s role narrows under an Opium Act amendment expected in 2027.
💼 Market — All 10 Wietexperiment Growers Now Active As Enecta Wins EU Catalogue Listing
The tenth and final cannabis experiment grower, Legacy Brands, began delivering on 2 March 2026, meaning all 10 licensed growers in the Dutch Wietexperiment are now active — a milestone that shifts the policy question from “can the supply chain be built?” to “does legal supply displace the illegal market?” The experiment is now fully operational after years of delays, licence disputes and build-out challenges, and sets the stage for the government’s evaluation ahead of the August 2028 deadline.
- The ramp-up accelerated in the final stretch: Growery (ninth grower) started supplying in November 2025, followed by Linsboer (eighth) shortly before. The first three growers — Fyta, CanAdelaar and Aardachtig — have been active since December 2023; Hollandse Hoogtes, Leli Holland, Holigram and Q-Farms joined through 2025.
Enecta‘s new Carmanecta hemp variety was approved for the Dutch national catalogue, triggering EU-level listing and clearing cross-border seed marketing across the bloc. The variety posted ~16.9 t/ha dry biomass yield in Raad voor Plantenrassen VCU trials, outpacing leading French, Ukrainian and Italian legacy fibre cultivars — planting seed is expected for the 2028 season.
- Village Farms International launched ten new cannabis products in the Netherlands, advancing product innovation in Europe’s largest adult-use programme.
🔬 Science — High-Fat Meal Boosts CBD Bioavailability 17-Fold
A Wageningen University crossover trial (n=11) found a high-fat meal increased CBD peak plasma concentration 17.4-fold (GMR 90% CI 12.4–24.2) and total exposure 9.7-fold (AUC 90% CI 7.7–12.3) from a broad-spectrum extract equivalent to 70 mg CBD — the largest food effect reported for any CBD formulation (Scientific Reports).
- A notable biphasic absorption pattern emerged under fed conditions, with a second plasma peak ~5 hours after the first — attributed to lymphatic transport and enterohepatic recirculation. The food effect on Cmax appeared more pronounced in males, though overall bioavailability was comparable across sexes.
- CHDR Leiden is recruiting patients for a placebo-controlled crossover trial of THC cannabis oil for chronic neuropathic pain, aiming to identify patient characteristics that predict analgesic response — a precision-medicine approach to cannabinoid prescribing.
- Grodan and Innexo demonstrated that eliminating the vegetative phase in cannabis cultivation boosts annualised yield 12% while cutting energy use 30% and labour 37%, with Grade A flower rising from 20% to 35% across six annual crop cycles versus four — a method the researchers expect to become the industry standard within five years.
🇮🇱 Israel — Court Blocks 165% Import Duty For 135,000 Patients As Licence Revocations Reach Supreme Court
🔭On the radar: Kolibri and Greencom Supreme Court appeals — enforcement deadline was 5 March 2026, outcome imminent.
🏛️ Regulation — 83 kg Inventory Gap Seals Greencom Revocation As Intelligence-Only Shutdowns Face Constitutional Test
Greencom, Trichom and Green Fields lost a petition to impose a 165% anti-dumping duty on Canadian imports, with the Jerusalem District Court ruling in favour of Finance Minister Bezalel Smotrich on macroeconomic grounds. The decision, backed by the Competition Authority, Government Legal Counsel and Ministry of Health, maintains duty-free imports for approximately 135,000 medical cannabis patients.
- The same court upheld permanent licence revocation for the Greencom group (Greencom Agro, The Technology Company, Greencom 4U) after intelligence indicated cannabis leakage to criminal organisations, including an unexplained 83 kg inventory gap from an August 2025 audit. Judge Eli Abarbanel also rejected a management-transfer rescue plan involving retired senior police officers.
- A ruling against the state could reshape enforcement across Israel’s ~135,000-patient medical cannabis sector, where intelligence-based revocations have become an increasingly common tool against suspected black-market leakage.
- Kolibri and Greencom filed urgent Supreme Court appeals on 25 February arguing ‘judicial blindness’ — no formal investigation, no cautioned questioning — challenging whether confidential intelligence alone can justify business closures. Both face a 5 March 2026 enforcement deadline. ⏳ Pending.
- The Jerusalem District Court separately rejected a petition to impose a 165% anti-dumping duty on Canadian cannabis imports, upholding Finance Minister Bezalel Smotrich‘s veto.
- The court found the proposed tariff would cause “certain price increase” for ~135,000 patients, harm product variety and constitute unjustified regulatory intervention given rising local production and record exports (~7.5 tonnes, Jan–Sep 2025).
- Petitioners Greencom, Trichom and Green Fields were ordered to pay NIS 40,000 in costs; the Competition Authority, Ministry of Health and Government Legal Counsel all opposed the duty.
💼 Market — InterCure Posts NIS 265M Revenue As Jane.co.il Launches Digital Marketplace
InterCure announced preliminary 2025 revenue of NIS 265 million alongside positive adjusted EBITDA and NIS 43 million in cash, signalling continued growth in Israel’s largest listed cannabis operator.
- Jane.co.il launched as a technology platform connecting patients, pharmacies and producers across Israel’s ILS 1 billion+ medical cannabis market with real-time pricing, CRM tools and demand analytics.
🔬 Science — Deep Learning Pipeline Achieves 98.6% Trichome Classification Across 15,000 Images
Ben-Gurion University and RCK Science-Based Cannabis Genetics developed a deep learning pipeline achieving 98.6% accuracy in classifying cannabis trichomes as clear, cloudy or amber from smartphone macro images. Across 15,000+ images and six varieties, the green-to-orange stigma colour transition aligned closely with peak cannabinoid concentrations — offering cultivators a scalable, low-cost harvest-timing tool.
- Innocan Pharma advanced its veterinary CBD pipeline: a liposomal CBD injection showed breakthrough pain relief in an elderly donkey with osteoarthritis, while LPT-CBD demonstrated efficacy over placebo in a randomised clinical study in dogs.
- Innocan secured its first patent in India for the liposomal CBD injection and received an FDA CVM sponsor fee waiver for the third consecutive year.
- A four-year study (n=241) at Rabin Medical Center found inhaled cannabis therapy associated with a 66% pain reduction (NRS 8.08→2.71) and 33% disability improvement over four years, with 81% retention. THC showed stronger analgesic effects (β=−0.15/g) than CBD (β=−0.08/g).
- An Israeli study found omega-3 fatty acid supplementation from late pregnancy to early lactation attenuated endocannabinoid system activation in preovulatory follicles and endometrium of Holstein dairy cows — linking cannabinoid science to agricultural reproduction.
🇨🇭 Switzerland — Grashaus Pilot Halves Illegal Market Use After Two Years
🔭On the radar: Federal Council CanPG revision in summer 2026; parliamentary debate on Cannabis Products Act in autumn 2026.
🏛️ Regulation — CanPG Consultation Splits Cantons As Zurich And Bern Reject Draft And Geneva Backs It
Two-year results from the Basel-Landschaft Grashaus Projects pilot showed legal cannabis shop access roughly halved illegal-market purchases — from ~20 to ~10 days/month — among over 1,600 participants. Sourcing from pilot shops surged from 6% to 84% within six months, providing the strongest empirical evidence yet that regulated retail displaces illicit supply.
- Operated by Sanity Group (now an Organigram subsidiary) under ISGF scientific oversight, the pilot also saw budtender trust as an information source jump from 53% to 79%. Participants shifted toward activated carbon filters and grew more open to edibles and vaporisers.
- The Federal Council received over 150 consultation submissions on the draft Cannabis Products Act (CanPG), with a narrow majority of cantons — including Zurich, Bern and Aargau — rejecting the draft as too complex and bureaucratic, while Geneva, Vaud and Basel-Stadt support the approach. ⏳ Federal Council revision summer 2026; parliamentary debate autumn 2026; potential referendum December 2028.
- SODK and GDK rejected the proposed steering levy in favour of a dedicated consumption tax; online commerce divided opinion between youth-protection concerns and black-market displacement arguments from FDP and GLP.
- EKSN and Addiction Switzerland endorsed the systemic change, stating prohibition has failed. SP, Greens and GLP back the reform; SVP, EVP and the Centre fundamentally reject it.
💼 Market — 34-Fold Pharmacy Cannabis Oil Price Variation Exposed
A saldo investigation found Swiss pharmacies charge vastly different prices for cannabis oil — from CHF 0.05/mg THC (mail-order via Avant-Care and Phyto-X-tract) to CHF 1.76 (Bahnhof-Apotheke Langnau, owned by Galenica) — a 34-fold spread that patients cannot compare in advance due to the narcotics advertising ban. An estimated 10,000 patients rely on medical cannabis, but only ~70 currently receive BAG health-insurance reimbursement, with monthly out-of-pocket costs reaching CHF 500.
- Addiction Suisse cross-referenced five data sources to profile Vaud’s illicit cannabis market: flower THC averages 13–15%, resin ~30% (up from 22% in 2023), with prices at ~10 CHF/g for flower and ~8 CHF/g for resin. Most Cann-L pilot participants nearly exited the black market after six months, with pilot prices (9–12 CHF/g) competitive at small quantities.
- Pure Holding AG positioned itself as a regulatory partner ahead of CanPG legalisation, leveraging its compliance track record since 2018 and founding membership of IG Hanf. Regulators from BAG and Swissmedic actively seek the company’s input on quality standards; new CEO Patrick Lämmli leads the strategy as ~700 CBD-era entrants have thinned.
🔬 Science — Altered Endocannabinoids In Adolescents With Major Depressive Disorder
A University Children’s Hospital Zurich–ETH case-control study (n=82 matched pairs) found lower plasma endocannabinoids (AEA, EPEA, DHEA) and reduced DHA- and AA-derived oxylipins in adolescents with major depressive disorder, supporting these lipid mediators’ role in pMDD pathophysiology. The Swiss National Science Foundation-funded study calls for intervention trials with omega-3 supplementation to test causality.
- A scoping review with Swiss institutional lead synthesised 53 studies on substance use and work ability in chronic pain patients, finding opioid use frequently associated with reduced work ability and increased absenteeism — highlighting integrative strategies as medical cannabis gains traction as an alternative.
🇲🇰 North Macedonia — 40 Tonnes Seized And 18 Licences Revoked As Sector Faces Integrity Crackdown
🔭On the radar: Ministry inspections of all 43 remaining licensed entities ongoing — 13 of 43 completed as of late February
🏛️ Regulation — Alphapharm And Five Others Named As Ministry Inspects All 43 Licensed Producers
North Macedonia launched its largest anti-drug operation in February, seizing over 40 tonnes of cannabis from companies licensed for medicinal cultivation — valued at €7–10 million — and revoking 18 of ~60 licences issued since the 2017 law. The crackdown, triggered by a cross-border trail to Serbia, exposed systemic diversion from the legal supply chain; PM Hristijan Mickoski warned seizures could reach 100 tonnes.
- Health Minister Azir Aliu revoked licences from six named companies — Alphapharm, Green Life, CBD Medplant, Life Plant, Atapharm and Cannabinoids — citing smuggling to Serbia, improper storage, inconsistent reporting and incomplete documentation.
- Alphapharm Group was directly implicated after inspections uncovered significant discrepancies between recorded and actual cannabis quantities on its premises.
- The Ministry of Health confirmed urgent inspections of all 43 remaining licensed entities, involving MALMED, the Agricultural Inspectorate, the State Institute for Health Protection and the Cannabis Commission; 13 inspected as of 24 February. ⏳ Inspections ongoing.
- Aliu announced new legislation aligned with EU directives will follow once inspections are complete, citing “weaknesses in the law” that enabled abuse.
- The operation began on 5 February after Serbian authorities seized ~5 tonnes of marijuana and weapons near Kruševac, reportedly originating from a Skopje-based factory licensed for medical cannabis production.
- Approximately 9 tonnes and 1,300 bottles of cannabis oil were seized from a Skopje company, with a further 31 tonnes confiscated from facilities in the eastern Strumica region.
- Mickoski blamed the previous SDSM–DUI coalition’s “cartel policy” for the licensing failures; the Organised Crime Prosecutor’s Office coordinates cross-border investigations with Serbian counterparts. ⏳ Court proceedings pending.
🇿🇦 South Africa — Draft Cannabis Act Regulations Set 750g Cap As Legal Analyses Expose Contradictions
🔭On the radar: DTIC draft commercial cannabis policy to Cabinet in April 2026.
🏛️ Regulation — 750g Fresh-Weight Cap And Five-Plant Limit Draw Legal Challenges Over Definitional Gaps
South Africa’s Department of Justice gazetted draft regulations under the Cannabis for Private Purposes Act on 3 February — 20 months after the Act was signed — proposing a 750g possession cap, a five-plant cultivation limit regardless of strain, and detailed transport concealment rules with an expungement process for prior convictions. Public comments closed 5 March 2026; proposed limits must still be submitted to Parliament for approval. ⏳ Parliamentary submission pending.
- SACHIDA welcomed the draft regulations as a milestone toward CPPA implementation but flagged definitional issues and personal possession levels, committing to submit formal representations by the 7 March 2026 deadline.
- Legal analyses exposed multiple contradictions in the draft rules. A detailed critique found the 750g cap internally inconsistent, questioned the five-plant limit’s adequacy, identified transport “deputisation” obligations on drivers, estimated ~1,100 cannabis clubs could form under the framework, and flagged gaps in the expungement mechanism.
- Adams & Adams highlighted the gap between harvested and dried cannabis weight — 750g of freshly harvested material yields far less when dried — arguing the regulations fail to account for the distinction, creating enforcement ambiguity.
- A separate analysis found the draft’s “permissible strains” restriction for transport lacks a national strain register, rendering the concept impractical and potentially unenforceable.
- The gazette specifies cannabis must be concealed in a boot or sealed container during transport, prohibits mixing with any other substance — a measure targeting nyaope — and bars handling or inspection in transit. International coverage framed the 750g limit as equivalent to ~2,000 joints, noting ambiguity over whether the cap applies per person or per shared private space.
- President Ramaphosa‘s SONA 2026 address on 13 February repeated an identical 14-word cannabis line from the previous year. The DTIC confirmed a draft commercial cannabis policy for Cabinet by April 2026 and an Omnibus Cannabis Bill targeting Parliament in 2027. ⏳ DTIC draft policy expected April 2026; Omnibus Bill targeted 2027.
🔬 Science — Review Links Phytocannabinoids To 76.9% Rise In Male Infertility Since 1990
A University of KwaZulu-Natal review of phytocannabinoids and the endocannabinoid system documented a 76.9% rise in male infertility since 1990 and found THC disrupts the hypothalamic-pituitary-gonadal axis — impairing sperm motility, morphology and testosterone levels — while CBD’s long-term reproductive effects remain uncertain.
🇫🇷 France — Health Authorities Set 2027 Generalisation Calendar As Reimbursement Framework Takes Shape
🔭On the radar: Permanent medical cannabis framework decree targeted June 2026.
🏛️ Regulation — DGS Presents Pricing Draft As Patient Associations Warn Conseil d’État Delays Threaten 3,000 Patients
Four regulatory milestones in rapid succession moved France’s medical cannabis experiment toward a permanent framework. The ANSM pilot — launched in March 2021 with approximately 3,000 patients — was extended into 2026 after the transition stalled, but decrees signed on 21 January 2026 were simultaneously transmitted to the Conseil d’État for validation.
- Four patient associations wrote to the Conseil d’État vice-president on 12 February urging rapid validation of the decrees, warning that delays threaten continuity of care for the 3,000 patients who have relied on the experiment since 2021.
- The DGS and DSS presented a pricing and reimbursement draft on 18 February, opening a 3-week to 1-month consultation. The UIVEC indicated reimbursement is expected but the pricing level will be decisive for commercial viability. Over 250 stakeholders attended Cannabis Europa Paris on 19 February.
- Health authorities subsequently set the formal calendar for generalisation of therapeutic cannabis by 2027 — the first time a concrete timeline has been attached to the transition from experiment to permanent framework. ⏳ Consultation ongoing; generalisation targeted 2027.
💼 Market — PGP Farmer Raises €3M For EU-GMP Site As Boiron Partners With Panaxia For 21-Country Push
PGP Farmer closed a €3M funding round to build an EU-GMP medical cannabis production site on a 9-hectare plot in France, targeting 30-tonne long-term capacity. The investment addresses a structural supply gap: approximately 75% of EU medical cannabis flowers are currently imported.
- Boiron signed a partnership with Panaxia Malta, with an option to acquire a majority stake by 2029 for €3.2M. The collaboration — rooted in both companies’ participation in the ANSM experiment — targets distribution across 21 EU countries.
- Danish producer Stenocare and ICMF submitted a regulatory dossier for the patented ASTRUM 10-10 oil to HAS for reimbursement evaluation, with Movianto appointed for nationwide pharmaceutical distribution. Independent analysts estimate ~300,000 patients could qualify under a permanent framework.
🔬 Science — OFDT Survey Shows Continued Decline In Cannabis Use Among French Secondary School Students
The OFDT EnCLASS 2024 survey (n=11,000) found cannabis experimentation among lycéens fell to 16.1% — down from 22.5% in 2022 — with all consumption indicators declining. The survey also measured synthetic cannabinoid use for the first time at 3.4%, establishing a baseline for monitoring novel psychoactive substance exposure among French secondary school students.
- 🇵🇫 The Louis Malardé Institute cultivated therapeutic cannabis in three controlled containers at its Paea facility, growing 13 plants under monitored temperature, humidity, CO₂ and LED light cycles.
- Head of natural substances Édouard Suhas noted Polynesia had no local CBD-producing varieties — only THC-rich pakalolo imported in the 1970s — requiring the institute to source seeds from approved international catalogues.
- The indoor trials demonstrated that THC content remained at or below the 0.3% legal threshold even after six successive transplants, validating the genetic stability of imported cultivars under Polynesian conditions. Flower analysis was conducted via chromatography at the institute’s Papeete laboratories.
- A retrospective study at a French multidisciplinary pain centre found a three-month dronabinol supply shortage (December 2023 – February 2024) caused significant clinical deterioration in chronic pain patients: 86% reported health worsening, permanent pain cases increased five-fold, and sleep quality declined significantly.
🇪🇺 EU — EFSA Sets Provisional CBD Intake At ≈2 mg/Day As EIHA Pushes 1% THC Under CAP 2028–2032
🏛️ Regulation — EFSA’s Conservative Threshold Challenges Most CBD Formats As Spain Restricts To Oral Solutions
EFSA established its first provisional safe intake level for CBD at approximately 2 mg/day for a 70 kg adult — applicable only to food supplements with ≥98% purity. Far below commercial dosing of 10–50+ mg, the threshold effectively challenges most CBD food and supplement formats on the European market; safety remains unestablished for under-25s, pregnant or lactating women, and those on medication.
- The EIHA acknowledged the provisional level but called it “particularly conservative” — significantly below the 17.5 mg/day supported by its own toxicological studies and the 10 mg/day applied in the United Kingdom. With a safe level now established, novel food authorisations can advance toward final approval; the association expects revision as additional data become available.
- Spain‘s AEMPS published formulary FN/2026/FMT/043, restricting medical cannabis to oral solutions for four indications — chronic pain, MS spasticity, epilepsy and chemotherapy-induced nausea — with a 32.4 mg THC/day cap. The decision mirrors France‘s existing flower exclusion, reinforcing a pan-European shift toward oral-only medical cannabis formats.
- The European Commission proposed recognising all parts of the hemp plant within the EU agricultural framework; the EIHA welcomed the move and called for a harmonised 1% THC cultivation threshold under the CAP 2028–2032 review, arguing the current 0.3% limit no longer reflects agronomic realities and exposes farmers to disproportionate sanctions from climate-related variability.
- EU Parliament Agriculture Committee discussions are expected to begin in March 2026, though some Member States are already resisting whole-flower authorisation. The EIHA benchmarks against Switzerland, Australia, New Zealand and South Africa to argue that 0.3% is agronomically outdated. ⏳ Parliament and Council debate expected spring–summer 2026.
🇵🇹 Portugal — Great Soul Pharma Loses Licence As Cultivation Sector Faces Credibility Test
🏛️ Regulation — 11 Pharmacy Preparations But Zero Subsidies Expose Access Gap
Portugal’s OPCM has authorised 11 pharmacy-compounded cannabis preparations, but none carries a public subsidy — leaving patients to bear the full cost while the industry channels production toward higher-margin EU export markets. The gap between export infrastructure and domestic patient access has become the defining tension in Portugal’s medical cannabis framework.
💼 Market — Licence Suspensions Continue As Iberfar Scales Eight-ACM Pipeline
Portugal’s cultivation sector faces a credibility test after Great Soul Pharma lost its production licence under Operation Ortiga — the country’s most significant cannabis enforcement action. CannaConnect moved to fill the resulting supply gap, while Iberfar‘s eight-ACM pharmaceutical pipeline and Cânhamor‘s €30 million hemp construction investment signal that serious capital is bifurcating away from cultivation risk toward downstream manufacturing and industrial applications.
- Great Soul Pharma had its production and commercialisation licences revoked by INFARMED following Operation Ortiga, in which the Judiciary Police investigated suspected criminal activity at the company’s facilities.
- Iberfar, part of the century-old Grupo Medinfar, outlined an integrated medical cannabis strategy spanning eight authorised cannabis-based medicines and five new formulations — including tablets, oral liquids and transdermal patches — for both domestic supply and EU export.
- Herbolea Biotech, Iberfar’s cannabis subsidiary, developed a GMP-certified solventless API through FAI Therapeutics, achieving ≥90% total cannabinoid content and >80% THC purity — positioning Portugal as a source for high-grade pharmaceutical intermediates.
- SOMAÍ partnered with Japan’s KISEKI to introduce Japanese-quality medicinal cannabis to global medical markets, leveraging SOMAÍ’s Portuguese EU-GMP production and distribution infrastructure.
- Cânhamor will begin exporting hemp ecoblocks to Germany through a partnership with Von Hanf, targeting capacity of 300 houses per month from an estimated €30 million investment — the largest hemp construction commitment in the Iberian market.
- The Pharma Science Global Conference, hosted by LEF, is scheduled for 12 March 2026 in Lisbon — one of the first major European pharmaceutical cannabis events of the year.
🔬 Science — Screen Systems Boost Iberian Poly Greenhouse Yields Up To 25%
A Wageningen University & Research study confirmed that screen systems increase crop yields up to 25% in Spanish and Portuguese poly greenhouses by boosting photosynthetically active radiation 30% and stabilising humidity and temperature. Van der Valk Horti Systems reported demand tripled year-on-year to 180 hectares of installations.
🇮🇹 Italy — Art. 18 Hemp Cases Favour Operators As OMC Export End Threatens Medical Supply
🏛️ Regulation — Courts Apply Offensività Principle In Hemp Seizures
Canapa Sativa Italia launched the Osservatorio Art. 18 — Italy’s first searchable legal case archive on industrial hemp seizures, acquittals and restitutions — documenting 15 cases across 8 of 20 regions since June 2025. Seven resulted in product restitution and 5 produced favourable outcomes (4 archiviazioni and 1 acquittal), with courts consistently applying the offensività principle, proportionality standards and technical evidence quality assessments.
- PazientiCannabis APS presented a technical report at a University of Messina roundtable exposing sharp access barriers in Sicily — the region reimburses only 3 of 6 nationally covered conditions, restricts prescribing to hospital specialists and caps treatment plans at 30 days.
- No digital prescription system exists; pharmacies dispensing medical cannabis remain concentrated in metropolitan areas, leaving rural patients underserved.
- The Dutch Office of Medicinal Cannabis (OMC) confirmed that 2026 will be its final year of cannabis exports, raising supply concerns for Italian patients dependent on Bedrocan phytocomplex products. Italian patient advocates warned the cessation could disrupt continuity of care, while Health Farm Srl positioned its domestic HF1 product as an alternative source.
- Enecta, whose Italian operations lost an estimated €150,000–€200,000 after the government’s 2025 CBD e-commerce shutdown, saw its new Carmanecta hemp variety win EU catalogue listing via the Dutch national register — a fibre cultivar posting ~16.9 t/ha dry biomass yield with potential to outpace Italy’s legacy Carmagnola.
💼 Market — MMP Becomes First Private GMP-Certified Lab As Tilray Expands Italian Operations
The OMC (Bureau voor Medicinale Cannabis) plans to end exports by 2026, threatening Italy’s Bedrocan-dependent medical supply chain. Italian pharmacies rely on Dutch-produced Bedrocan varieties; the OMC confirmed 2026 will be its last export year as the Netherlands restructures its medical cannabis framework, with commercial companies expected to take over.
- The SCFM confirmed a production quota of 400 kg for 2026 — unchanged since 2022 and down from 500 kg in 2021 — well below estimated demand, underscoring Italy’s structural dependency on imported supply as the OMC exit looms.
- With over 50,000 patients in therapy and sales up 25% YoY, domestic production covers less than 20% of the estimated 1,200 kg demand; the SCFM remains capped at 400 kg/yr and 2024 total supplies reached 1,694 kg.
- Materia Medica Processing (MMP), a Siena-based startup within Toscana Life Sciences, obtained AIFA and UCS authorisations to become Italy’s first private GMP-certified cannabis pharmaceutical lab, covering processing, distribution and packaging of cannabinoid-based APIs.
- MMP can import, analyse and distribute medical cannabis inflorescences — including the Bedrocan variety — and balanced THC/CBD oils.
- Tilray Brands launched Tilray Medical Italia, rebranding FL Group to expand its authorised portfolio of flowers and oils available by prescription through Italian pharmacies.
- The company earlier formed a strategic partnership to broaden access to medical cannabis extracts.
- Over 100,000 prescriptions were issued between 2019 and 2024, reaching approximately 28,000 patients — average age 60, 2.3:1 female-to-male ratio and 78% for chronic pain. Experts called for a legislative review a decade after the framework law.
- CLINN launched Italy’s first patient protection service for cannabis treatment, having assisted over 8,000 patients with more than 15,000 consultations.
- Avextra is supporting the NEUROBIS Phase II clinical trial on cannabis-based medicines for neurodegenerative diseases in Italy — a 36-month, 180-patient randomised, placebo-controlled study authorised and funded by the Italian Ministry of Health, covering ALS, Parkinson’s and Alzheimer’s disease.
- CS MEDICA closed a 1.1 MDKK white-label CBD-infused product order for Italy, targeting MDR Class IIa products for distribution in over 50 countries.
🇹🇭 Thailand — 7,000+ Shops Close As Election Cements Medical-Only Controlled-Herb Regime
🏛️ Regulation — Only 15.5% Of Licences Renew As All Major Parties Back Decriminalisation Reversal
Thailand’s 8 February 2026 national election confirmed broad political consensus for a medical-only cannabis framework, with most parties explicitly backing the reversal of the 2022 decriminalisation experiment. The vote consolidates the June 2025 reclassification of cannabis flower as a “controlled herb”, which mandated prescriptions and compliance obligations for dispensaries after widespread recreational retail fuelled rising hospitalisations and youth mental health pressure.
- The controlled-herb regime requires prescriptions from licensed medical practitioners, imposes record-keeping obligations on dispensaries and mandates compliance standards that most retail outlets — opened during the rapid 2022–2025 expansion — could not meet. Rising public health concerns including increased hospitalisations and poison-centre exposures, particularly affecting youth, provided the political justification for reimposing controls.
- The IDPC noted contested cannabis policy as a defining election issue, with most parties favouring a medical-only framework and a potential full reversal of the 2022 decriminalisation on the agenda.
- Though Bhumjaithai leader Anutin Charnvirakul — who championed the original decriminalisation — stayed on as prime minister, he has reframed the policy as always focused on medicinal use.
- Surveys show the overwhelming majority of Thais oppose recreational legalisation; even tourism bodies support reclassification after losing high-value family holidaymakers.
💼 Market — Bangkok Shops Resort To Telehealth Prescriptions As Tourism Stays Below Pre-COVID Levels
Thailand’s cannabis industry faces continued uncertainty after the Bhumjaithai Party‘s snap election victory on 8 February, with ~18,000 dispensaries that opened after 2022 decriminalisation decimated by the prescription-only regime.
- Surviving Bangkok shops resort to telehealth prescription workarounds — including A-frame signs advertising “free prescription with every purchase” — while tourism (~15% of GDP) remains below pre-COVID levels.
- California-based Sunderstorm began distributing Thai-grown cannabis flower into the UK, Germany and Australia through its Kanha brand and Thai Stick Holdings JV, with Grow Group UK describing it as the first premium Thai-grown flower in the UK market — signalling Thailand’s emergence as a cannabis flower origin country for regulated international markets despite the domestic policy reversal.
🔬 Science — Cannabis Oil Matches Lorazepam In Insomnia RCT; Portable THC Gas Sensor Developed
A three-arm open-label RCT in Kamphaeng Phet Province (n=60) found Cannabis sativa oil comparable to lorazepam for chronic insomnia over four weeks: PSQI scores fell from 13.6 to 5.1 (p<0.001) versus 14.4 to 5.8 for lorazepam 1 mg. Quality-of-life gains were markedly larger in both plant-based arms (EQ-VAS ~50→82) than the benzodiazepine control (50.8→59.2), with only mild adverse reactions in 5% of cannabis oil participants.
- Researchers at Chulalongkorn and Kasetsart universities developed the first electrochemical gas sensor for Δ9-THC in cannabis smoke, achieving a 4.5 ppb detection limit on a disposable MnO₂-modified graphene electrode validated against HPLC and GC–MS — a low-cost, portable tool for air-quality monitoring in cannabis consumption areas (Analytical Chemistry).
🇲🇦 Morocco — ANRAC Authorises First CBD Medicine As Cannablanca Builds 145M MAD Pharma Unit
🔭On the radar: Cooperative-model planting season outcomes and new seed rule implementation.
🏛️ Regulation — 116 Products Registered Across Five Medical Specialties As Clinical Trial Launches At CHU Tanger
ANRAC reported significant progress in Morocco’s therapeutic cannabis programme, with 116 products registered — 56 food supplements, 59 cosmetics and the first locally manufactured CBD medicine for refractory childhood epilepsy, which received marketing authorisation.
- A clinical trial is under way at CHU de Tanger to evaluate CBD efficacy, with over 600 authorised sales points and five validated medical specialties: internal medicine, neurology, dermatology, geriatrics and paediatric gastroenterology. ⏳ Oncology, psychiatry, rheumatology and pain/anaesthesia-resuscitation under validation.
- The AMCUC urged acceleration of the clinical phase for therapeutic cannabis, highlighting the impact of regulatory delays on medical practitioners and patients.
💼 Market — Chefchaouen Pharma Unit 80% Complete As $260M Sector Reaches 2,647 Licensed Farmers
Cannablanca is building Morocco’s first cannabis pharmaceutical unit in Chefchaouen — a 145M MAD investment, 80% complete, targeting 200–300 jobs and pharma-grade exports. The project marks Morocco’s transition from biomass supplier to pharmaceutical producer as the legal cannabis sector was valued at $260.3M with 2,647 licensed farmers, 2,169 ha cultivated and 4,082 tonnes of biomass.
- Swiss export prices reached €1,400–1,800/kg; Pakistan was reported as following Morocco’s regulatory model for its own cannabis programme.
- 109 CBD-based products (50 dietary supplements, 50 cosmetics) were on the market through 600+ retail outlets — figures subsequently updated to 116 products by ANRAC‘s February disclosure.
🔬 Science — Urology Congress Reports ~65% Pain Reduction As Hassan II Screens Cannabis For Skin Cancer
Morocco’s 14th urology surgery congress highlighted medical cannabis as a promising chronic pain treatment, with Carola Pérez (Spanish Observatory for Medical Cannabis, IACM) presenting data showing ~65% pain reduction, 58% fewer hospital visits, 55% fewer GP consultations and 65% lower medication consumption.
- Hassan II University of Casablanca screened 49 cannabis phytoconstituents for skin cancer targets, identifying compounds C5, C6 and C7 as promising multi-target inhibitors of EGFR, BRAF V600E and TGF-β via integrated computational investigation.
- ANRAC launched a 10-month hemp feed study for the poultry sector — the first systematic evaluation of cannabis-derived feed formulations in Morocco.
- A Marrakesh study of 95 cannabis users reported high prevalence of visual symptoms: 75.8% red eyes, 67.4% ptosis and 66.3% light sensitivity.
🇨🇴 Colombia — Invima Faces March Deadline As Decreto 1138 Reshapes Medical Cannabis Framework
🔭On the radar: Flower launch in domestic pharmacies; certificate and THC threshold harmonisation for cross-border trade.
🏛️ Regulation — Invima Must Issue Quality And Traceability Standards Under Decreto 1138 By March
Invima faces a March 2026 deadline to issue technical regulations under Decreto 1138 de 2025, establishing mandatory quality, traceability, and dispensing standards for medical cannabis. The package includes analytical standards for THC and CBD, good manufacturing practices for magistral preparations, and interoperability with the SEED platform for end-to-end traceability from cultivation to patient. ⏳ Regulations expected March 2026.
- Carmen’s Medicinals CEO Juan Romero said the strengthened controls will enable medical cannabis to be perceived as a scientifically backed treatment; the Ministry of Health will update prescribing and safe-use protocols in parallel, reducing legal uncertainty for practitioners.
- Senate candidate Marcela Fúquene launched a campaign under the Fuerza Ciudadana coalition with regulated cannabis as a central policy plank, participating in the SINTRABACANN debate — signalling growing mainstream political traction ahead of legislative elections.
💼 Market — Green Marketplace Opens Colombia’s First Cannabis Pharmacy As Greenlab Ships 150 kg To Europe
Green Marketplace opened Colombia’s first specialised cannabis pharmacy in Medellín, backed by SENA Antioquia — which has trained over 1,000 people in cannabis-sector skills since 2022. The pharmacy is the first retail dispensing point in a domestic market that has been almost entirely export-oriented, signalling a structural shift toward local patient access.
- Greenlab exported 150 kg of cannabis flower to Europe in recurrent shipments, while Earth’s Healing Colombia convened an export synergies roundtable in Cali targeting licensed producers and GACP-certified firms — evidence of a maturing export corridor.
- PharmaCielo (TSXV: PCLO) announced its AGM for 2 April 2026 alongside an intention to issue ~12.2 million shares at C$0.08 to cover C$972,317 in accrued debenture interest — a debt-for-equity conversion signalling continued capital constraints.
🔬 Science — Biostimulant Field Trials At Clever Leaves And Postoperative Pain Meta-Analysis
Biotech Tricopharming Research‘s novel Tricostimulant™ botanical biostimulant enhanced cannabinoid content in field trials across three sites in Spain and Colombia — including Clever Leaves‘ Tocancipá facility (Ecomedics S.A.S.) — boosting CBD-A by 32%, CBG by 48% and CBN by 20% in the Carmagnola Selezionata variety (International Journal of Plant Biology, 2026, 17(3)).
- A systematic review and meta-analysis of cannabinoids for postoperative pain synthesised clinical trial evidence on efficacy and tolerability — a reference as Colombia’s medical cannabis prescribing framework evolves (European Journal of Pain / Wiley).
- The Universidad Nacional de Colombia opened a specialised course on dried flower production — eight sessions, 30 places, starting 11 April 2026 — led by Professor Javier Hidalgo, who has registered 54 cannabis varieties with the ICA and trained approximately 1,000 people since 2017.
🇵🇱 Poland — 5,450 kg Imported, €60M Market As ODI Pharma Turns Profitable
🏛️ Regulation — Chief Pharmaceutical Inspector Mandates Compounding Rules As Prosecutor General Backs Drug Threshold Table
Poland’s Chief Pharmaceutical Inspector (GIF) Łukasz Pietrzak ruled on 4 February that medical cannabis must be compounded as a lek recepturowy — quality-controlled, weighed, and labelled in pharmacy packaging — making dispensing in the manufacturer’s original container a legal violation, standardising inspections by Provincial Pharmaceutical Inspectors across Poland. The ruling simultaneously ended chronic patient–pharmacy conflicts: a valid prescription is sufficient for dispensing; pharmacists may not demand additional documentation or invoke personal objection.
- Poland’s Prosecutor General approved development of a drug threshold table defining “insignificant quantity” under Article 62a of the Act on Counteracting Drug Addiction — targeting stark regional disparities (2 g dismissed in Warsaw vs. 0.5 g prosecuted in Podkarpacie). An expert team of prosecutors, police officers and toxicologists will create binding values, with the Institute of Justice (IWS) collecting comparative data from European countries. ⏳ In progress; no implementation date set.
- The Polish cannabis patient association met with the Patient Rights Ombudsman on 6 February to raise concerns about telemedicine prescribing restrictions — signalling that access constraints remain a live political issue as GIF enforcement rolls out.
💼 Market — Post-Ban Volume Recovery Masks 19% Revenue Decline On Price Compression
Poland’s medical cannabis market dispensed 5,450 kilograms in 2025, confirming full volume recovery from the 2023 telemedicine prescription ban — yet revenue fell 19% to approximately €60 million (~253 million PLN) as average prices dropped 28% year on year. The divergence signals a market where access has normalised but operator margins are under structural compression.
- ODI Pharma reported H1 2025/2026 net sales of SEK 16.9 million and a profit of SEK 1.3 million, returning to profitability on Q2 revenues of SEK 10.9 million (+84.3% quarter on quarter) — a reverse profit warning reflecting strengthened delivery predictability in the Polish market.
- S-Lab cut the wholesale price for its Lilac Diesel variety (THC 18%), bringing the patient price to 300–330 PLN per 15 g depending on expiry date — a concrete signal of the downward pricing trend visible in the aggregate data.
- New strain launches continued to diversify pharmacy supply through February:
- Tilray listed Cataract Kush, an indica-dominant strain (THC ~22%) targeting chronic pain and insomnia patients, sold in 15 g sealed containers.
- Aurora announced Electric Honeydew (THC 29%, sativa-dominant), sharing the same Cannabis flos Aurora register as Sourdough — distinguishable only by batch code (EHD vs SRD), a packaging nuance patients must track at dispensing.
🔬 Science — Poznań Researchers Extend Berry Shelf Life With Cannabis-Extract Polysaccharide Films
Poznań University researchers developed polysaccharide films with Cannabis sativa extract that extended freeze-dried berry shelf life, reducing moulds and yeasts by 0.22 log cfu/g after eight weeks while improving film elongation from 32.5% to 44.8% with increased hemp extract concentration (Molecules, January 2026).
- A validated HPLC-DAD method for CBD determination in intravenous nanoemulsions based on CBD-enriched hemp oil advanced a delivery pathway for poorly water-soluble cannabinoids — relevant as clinical interest in injectable CBD formulations grows (Molecules / MDPI).
- A Food Chemistry study examined CBD transformation during high-temperature extraction in model systems, hemp and functional foods, highlighting thermal degradation risks for manufacturers using heat-based isolation methods.
- An MDPI Sustainability study tested five hemp fibre types for sound absorption across thickness (20–60 mm) and density (50–250 kg/m³) ranges using impedance-tube and Delany–Bazley–Miki modelling, finding hemp fibre effective as a sustainable alternative to conventional acoustic insulation.
- A review with Polish institutional lead surveyed wastewater-based epidemiology methods for synthetic cannabinoids, highlighting analytical challenges at ultra-trace levels and calling for larger, standardised EU monitoring campaigns (Molecules, January 2026).
🇪🇸 Spain — AEMPS Authorises Medical Cannabis For Four Indications But Zero Products Submitted
🏛️ Regulation — Oral-Only Formulary Caps THC At 32.4 mg/Day As Health Ministry Opens Door To Community Pharmacies
Two months after AEMPS authorised medical cannabis for four indications with an oral-only formulary capped at 32.4 mg THC/day, no operator has submitted a product — leaving the programme without a single available medicine despite 12 licensed cultivators and a complete regulatory framework.
- Secretary of State for Health Javier Padilla said evidence will determine dispensing flexibility: the decree centres on hospital pharmacies with specialist prescribing but allows autonomous communities to adopt alternative arrangements. ⏳ Decree review expected end-2026.
- The Health Ministry opened the door to dispensing in community pharmacies, a shift from the hospital-only model that would broaden access through the existing retail pharmacy network.
- The oral-only restriction mirrors France‘s flower exclusion; Business of Cannabis noted the pan-European shift away from flower raises questions about flower-based prescribing, with Curaleaf International CEO quoted on cross-border supply implications.
- The Health Ministry opened the door to dispensing in community pharmacies, a shift from the hospital-only model that would broaden access through the existing retail pharmacy network.
- AEMPS authorised 12 companies for cannabis cultivation as of January 2026 — spanning research, medical production and manufacturing process validation — establishing upstream supply capacity ahead of product submissions.
💼 Market — Ondara Secures 9,000 kg Contract As Quorum Receives Cultivation Licence
Ondara signed a 9,000 kg annual supply contract with one of the world’s largest cannabis companies, positioning the Soria-based producer among Europe’s reference suppliers. AEMPS separately authorised new high-demand genetics targeting key European markets, but critical cash-flow pressure means Ondara urgently needs Iberaval working-capital guarantees to execute the deal. ⏳ Financing pending.
- David Engel has injected >€1.8M since productive operations began in July 2025, reinstated all ERTE workers and formalised 12 new hires. Institutional support from the Junta de Castilla y León was acknowledged as decisive.
- Quorum Biomedical received its third consecutive AEMPS cultivation licence renewal for research-grade Cannabis sativa L. and announced a collaboration with Basque Country-based Sovereign Genetics for plant variety development.
- Spain’s AEMPS formulary restricts medical cannabis to oral solutions for four indications — mirroring France‘s flower exclusion — with a 32.4 mg THC/day cap, reinforcing a pan-European shift away from flower-based prescribing. (Cross-ref: EU section.)
🔬 Science — BIORCAN Invests €1M In Cosmetic Cannabinoid R&D
The BIORCAN project is investing €1M in cosmetic cannabinoid R&D, with partners Votum World, the University of Murcia, UCAM, Cetem and Global Biotech allocating 93% of funds to direct research costs — a significant Spanish commitment to CBD/hemp-derived cosmetics amid the ongoing ECHA reprotoxic review timeline for CBD in cosmetic applications across the EU.
- A socio-legal study published in International Journal of Drug Policy analysed how Spanish cannabis social clubs expanded through decentralised governance — particularly in Catalonia and the Basque Country — before Supreme Court and Constitutional Court rulings from 2015 onward curtailed the legal basis, reaffirming centralised authority over criminal law. The Basque model stayed closer to cooperative principles; Catalonia’s commercialisation triggered the crackdown.
🇨🇿 Czech Republic — F1 Hybrids Hit Record Yields As Labs Struggle To Identify Post-Ban Synthetics
🔬 Science — 49 Natural And 27 Synthetic Cannabinoids — And Labs Still Can’t Keep Up
Czech F1 hybrid cannabis varieties achieved record yields in controlled tests, signalling that next-generation genetics are reaching commercial viability for Central European cultivators across both recreational and medical supply chains.
- Analytical chemists at the University of Chemical Technology Prague (VŠCHT) detailed the growing challenge of testing cannabis products as synthetic compounds proliferate post-HHC ban. The ISO 17025-accredited lab quantifies 49 natural and 27 synthetic cannabinoids, but new unidentified substances are entering the Czech market with no published safety data.
- Identically labelled products contained different active ingredients and dosages; trace THC appeared in virtually all samples including synthetic CBD. In 2018, synthetic cannabinoids caused dozens of poisonings and two deaths in Czechia — a precedent researchers cite as the unregulated segment scales.
- A comprehensive Phytochemistry Reviews direction review concluded that cannabis liberalisation “seems inevitable”, mapping the research frontier across phytochemistry, pharmacology and regulatory science for policymakers, cultivators and patients.
🇩🇰 Denmark — First Medical Cannabis Social Club As STENOCARE Surges 215%
🏛️ Regulation — Smokenhagen Opens Copenhagen’s Prescription-Only Consumption Space
Smokenhagen, Denmark’s first cannabis social club restricted to prescription holders, opened in Copenhagen on 2 February 2026 — a members-only space for on-site consumption of pharmacy-dispensed cannabis under the country’s now-permanent medical framework, with approximately 1,800 patients prescribed since 2018 and around 20,000 prescriptions issued in total.
💼 Market — STENOCARE Surges 215% With CannGros Acquisition
STENOCARE reported FY 2025 net sales of DKK 7.02M (+215% YoY) with three consecutive quarters of positive EBITDA, driven by the November 2025 acquisition of CannGros — the leading Danish flower supplier — which is expected to add DKK 4–6M in annual revenue and support a 2026 break-even target.
- Q4 net sales reached DKK 2.6M (+107% YoY, +36% QoQ). Strategic partnerships secured with WEECO Pharma for an Astrum oil launch in Germany (February 2026) and first-mover distribution filing in France. ⏳ France partnership pending.
🔬 Science — THC Analog–CB1 Binding Mapped As CS Medica Secures Fourth EU Patent
A Nature Communications study elucidated the structural basis of THC analog activity at the CB1 receptor, mapping spatiotemporal ligand interactions critical for potency, efficacy and off-rates — advancing the mechanistic toolkit for cannabinoid drug design.
- CS Medica achieved its fourth European patent with the MDR-approved Cannasen psoriasis gel, extending its CBD-derived dermatology IP portfolio.
🇳🇴 Norway — Patient Access Shrinks 30% As Groups Demand Pilot Programme
🏛️ Regulation — Four Patient Groups Call For National Registry, Reimbursement And Driving Rules
A coalition of patient groups — Stopp Lidelsen, SMOFA, LARS and Ryggforeningen i Norge — published an open letter in Dagens Medisin demanding a national pilot programme for medical cannabis, citing a ~30% drop in patients receiving cannabinoid treatment since 2022 after funding shifted from national insurance (folketrygden) to hospital budgets. The authors propose a national registry study, prescribing guidelines, a reimbursement scheme and harmonised driving rules — noting Denmark made its eight-year pilot permanent and 21 EU states have integrated medical cannabis. ⚠️ COI: co-author Mikael Soedergren (Sapphire Nordics / Imperial College London) is medical director at Curaleaf International.
🇮🇪 Ireland — CannEpil Reimbursement Expands Paediatric Access As Neurologists Flag Prescribing Barriers
💼 Market — 1,000-Unit CannEpil Batch Ships Under Full Irish Reimbursement
Argent BioPharma shipped 1,000 units of CannEpil to Ireland — its largest single commercial batch — fully reimbursed under the Irish national health scheme at €467 per unit (~A$783,000 retail value).
- Distributed by Georgelle Pharma via Versa Pharmaceuticals, the cannabinoid formulation targets refractory epilepsy, where ~30% of patients fail standard therapies.
🔬 Science — Neurologists Survey Identifies Education Gaps In Cannabis Prescribing
An all-Ireland survey of neurologists — the prescriber group at the forefront of cannabis-based therapy in Ireland — found participants rated their knowledge as average and reached consensus that the application process for accessing cannabis-based medicines is unclear (Irish Journal of Medical Science). Approximately one-third had applied on behalf of a patient; the study calls for targeted educational programmes.
🇧🇪 Belgium — European-First Hemp Pilot Targets Belgium’s €330 Billion PFAS Crisis
🔬 Science — Ghent Port Greenhouse Tests Hemp Extraction On 100 m³ Contaminated Soil
OVMB (Eiffage Construction), C-biotech and C-ground (Cordeel Group) are piloting industrial hemp phytoremediation in a closed 100 m² greenhouse at Ghent port — a European first — to accelerate PFAS extraction from 100 m³ of contaminated soil without destroying the land. Belgium ranks as Europe’s worst PFAS hotspot, with 100% of the population in a risk zone and remediation costs estimated at a minimum of €330 billion; an EC study projected EU-wide PFAS costs at €440 billion by 2050.
🇦🇹 Austria — Review Warns Flowers, Extracts And Isolates Are Not Interchangeable
🔬 Science — Mislabelling And Undeclared THC Flagged Across OTC CBD Products
A review in MDPI Psychoactives argues that cannabis flowers, extracts and isolate cannabinoids are not interchangeable despite common treatment as equivalent, calling for detailed phytochemical disclosure beyond THC/CBD in both research and consumer products. The paper flags widespread mislabelling and contamination in OTC CBD products — including undeclared THC, pesticides and heavy metals — and highlights risks from semi-synthetic cannabinoids (Δ8-THC, HHC) entering the market with inadequate characterisation.
🇬🇷 Greece — Three Novel THC Analogs Identified In European Market
🔬 Science — CB9, Tresconol And CBx Reach Consumers Ahead Of NPS Frameworks
Researchers identified three novel THC analogs in the European market — CB9 ([2-(E)-propen-1-yl]-Δ8-THC-acetate), tresconol ([2-propen-2-yl]-Δ9-THC) and CBx — through structural characterisation of products marketed under novel cannabinoid branding (Drug Testing and Analysis / Wiley). The findings flag an expanding landscape of semi-synthetic THC derivatives reaching European consumers ahead of regulatory frameworks for novel psychoactive substances.
🇲🇹 Malta — Cannabis Club Stock Limits Tiered, Penalty Raised To €50,000
🏛️ Regulation — Clubs Tiered From 350 g To 3.5 kg; Maximum Fine Quintupled
Malta introduced tiered stock limits for cannabis clubs based on membership size, replacing the previous flat 500 g cap. Clubs with over 350 members may now hold up to 3.5 kg; smaller tiers scale down to 350 g for associations with up to 50 members. The maximum penalty for regulatory breaches was raised from €10,000 to €50,000 or twice the revenue from the offence, alongside clearer rules on due diligence and internal registers.
- Releaf Malta condemned the ARUC for sending enforcement officials to private homes to investigate 140 cannabis smell complaints received via freephone, calling it a privacy violation that reinforces stigma against consumers. ARUC chairperson Joey Reno Vella said most complaints were resolved after officials recommended actions to remove the inconvenience.
- Drug policy researcher Karen Mamo critiqued Malta’s reliance on deterrence-based drug policy, citing General Population Survey data showing cannabis last-month prevalence at 3.2% (up from 0.5% in 2001) and noting Malta is the only EU member state that does not share annual drug consumption data with the EUDA. The National Addiction Advisory Board lacks human rights, LGBTIQ or drug-user representation.
🇺🇾 Uruguay — Sales Reach 4,290 kg (+34%) As IRCCA Eyes Tourist Access
🏛️ Regulation — IRCCA Seeks Legal Pathway For Tourist And Foreign Worker Sales
The IRCCA is developing a mechanism to allow tourists, students and foreign workers to purchase regulated cannabis under the same conditions as registered residents, arguing that excluding temporary visitors channels them toward the illicit market. Director Martín Rodríguez confirmed the proposal; the agency’s annual medicinal report documented licence and product registration activity across 2025.
- A Udelar study found higher-THC medical cannabis registration is slower, concluding that Uruguay is “amputating” its ability to sell more psychoactive medicinal products — highlighting regulatory friction that limits both patient access and cultivator operations.
💼 Market — Pharmacy Prices Rise Across All Four Varieties As Four New Licences Awarded
Pharmacy sales reached a record 4,290 kg in 2025 (+34% vs 2024’s 3,207 kg), driven by the addition of higher-THC variants. The IRCCA has awarded four new production licences to meet growing demand, though current supply from three producers remains insufficient. The registered buyer base has reached 85,000 across 57 authorised pharmacies.
- Adult-use cannabis pharmacy prices rose by UYU 5 across all four varieties (Alfa, Beta, Gamma, Épsilon) effective 1 February 2026, with the cheapest now at UYU 485 and the most expensive at UYU 615.
- GreenMed, a Uruguayan cannabis laboratory, is positioning for global expansion in pharmaceutical cannabis products, investing in R&D and regulatory compliance from its Uruguay base — leveraging the country’s early-mover regulatory framework.
🇦🇷 Argentina — REPROCANN Automates Approvals For 240,000 Users
🏛️ Regulation — REPROCANN Automates Low-Risk Approvals For 240,000 Registered Users
SEDRONAR and the Ministry of Health launched automated digital evaluation for low-risk REPROCANN self-cultivation applications, targeting wait times that exceeded six months for approximately 240,000 registered users. Under-21 patients, mental health diagnoses and ONG/solidary grower requests remain under manual Medical Commission review.
- The automation follows Decreto 27/2026, which transferred the cannabis programme to SEDRONAR under a broader Ministry of Health restructuring — a 45.7% reduction in ministerial hierarchy — strengthening traceability and the therapeutic focus under Ley 27.350.
- CAINCCA president Luis Osler welcomed the measure as aligned with Resolución 1780, calling it “an indispensable tool for the entire system to function.”
💼 Market — Seed Bank Donates 400 Medicinal Seeds As CUMI Launches Hemp Food Production
Argentina’s cannabis grassroots and early-stage hemp sector advanced in parallel. A Radio Sativa industry outlook framed five themes shaping 2026 — regulation, market development, cultivation, manufacturing and investment — as operational developments confirmed bottom-up momentum in both medical and industrial verticals.
- Semas Argentinas donated 400 Moby Lemon seeds to vetted NGOs treating refractory epilepsy, autism and oncological pain — distributed exclusively to solidarity cultivation groups producing free medicinal oil.
- CUMI, a family project of five engineers, launched the first hemp genetics adapted to extreme heat in Santiago del Estero (4 ha scaling to 200–500 ha via smallholder integration), producing protein flour, cold-pressed oil and construction biomaterials with UNSE, CONICET, INTI and INTA.
🔬 Science — UNAJ And CONICET Launch Hemp Field Trial In Buenos Aires Horticultural Belt
UNAJ, CONICET and the Buenos Aires Ministry of Agrarian Development launched hemp field trials at Chacra Experimental El Pato in Berazategui, testing three sowing windows (20 October, 29 December 2025 and 19 January 2026) and crop association with tomato, squash and maize to evaluate biomass, grain yield per m² and phenological response across the horticultural belt.
🇲🇽 Mexico — Supreme Court Ends Automatic Trafficking Presumption Above 5 g
🏛️ Regulation — 14.26 g Case Shifts Burden Of Proof But Falls One Vote Short Of Unconstitutionality
Mexico‘s Supreme Court (SCJN) ruled that exceeding the 5g cannabis threshold can no longer automatically presume trafficking — authorities must now assess objective and subjective context case by case. The 5g limit remains in the General Health Law as an indicative reference, but the ruling, arising from a case involving 14.26g found on a person experiencing homelessness, fell one vote short of a broader unconstitutionality declaration.
- PRI deputy Mario Zamora Gastélum proposed legalising regulated medical opioid and CBD production in Sinaloa, aiming to transform the region into a pharmaceutical hub with full traceability and sanitary oversight — replacing illicit economies with formal employment and international supply-chain integration.
🔬 Science — Municipal Education Levels Predict Cannabis Use — Effect Nearly Doubles By 2023
An analysis of two nationally representative Mexican surveys (International Journal of Drug Policy) found municipal education levels positively associated with recreational cannabis use prevalence — a 1.5% increase per unit in 2016–17 rising to 2.9% in 2023, suggesting socioeconomic environment is an increasingly significant predictor of use patterns.
🇨🇱 Chile — Fifty Parliamentarians Petition Boric To Legalise Home Cultivation
🏛️ Regulation — Cross-Party Bill Proposes 6 Plants, 40 g Possession And 500-Member Social Clubs
A cross-party bloc of 50 deputies, led by Ana María Gazmuri (Acción Humanista), filed a bill to reform Law 20,000 — proposing legalisation of home cultivation (up to 6 plants), personal possession of 40 g, annual retention of 500 g, and non-profit cannabis social clubs of up to 500 members. The bill now enters the Comisión de Salud, then the Chamber floor and the Senado. ⏳ Committee stage; no scheduled date.
🇨🇷 Costa Rica — Licensed Farm Raided As US $100M Court Dispute Unfolds
💼 Market — Environmental Prosecutor Targets Coris Wetland Damage At Vantage Point’s Export Farm
Costa Rica’s Environmental Prosecutor raided Hybrida Farms — one of the country’s first licensed medical-cannabis export operations — over alleged damage to the Coris Wetland. The farm’s US parent, Vantage Point Global Corp., faces a US $100M fraud and conspiracy lawsuit in Travis County filed by former consultant Craig Aumann; a hearing on a temporary restraining order is set for next week. ⏳ TRO hearing pending; environmental investigation at confidential stage.
🇵🇾 Paraguay — Dinavisa Tightens Medical Cannabis Dispensing Rules
🏛️ Regulation — Resolution 488/2025 Channels Dispensing Through Authorised Pharmacies
Dinavisa published Resolution 488/2025 on 4 February 2026, tightening medical cannabis dispensing by restricting supply to authorised pharmacies and dispensaries — formalising pharmacy-based distribution within Paraguay’s medical cannabis programme.
🇯🇲 Jamaica — Jacana Completes First Regulated Cannabis Export
💼 Market — Oil, Vape And Extract Shipment Opens Caribbean Prescription Channel
Jacana completed Jamaica’s first regulated medical cannabis export to the Cayman Islands on 7 January 2026, shipping oil-based products, vape pens and extracts under permits from the Cannabis Licensing Authority and Cayman’s chief medical officer. Caribbean Medical Distributors will dispense via prescription through pharmacies and hospitals — a proof-of-concept milestone as the company, which operates a 100-acre farm and employs ~110 workers, explores European markets including Germany.
🇳🇿 New Zealand — Native Plant With Cannabis-Like Properties Discovered
🔬 Science —Endometriosis Pain Falls 31% Over 12 Weeks In First NZ Prospective Cohort
The first prospective cohort study of medicinal cannabis for endometriosis in Aotearoa (n = 28 completers) found overall pain fell from 5.46 to 3.77 and EHP-30 quality-of-life scores halved (68.77 → 37.40) over 12 weeks, with limited adverse events. University of Otago researchers called for larger controlled trials; qualitative interviews highlighted cost and driving restrictions as primary barriers to access.
- An iwi-linked discovery identified a tiny native plant with cannabis-like properties that Rua Biosciences is exploring for homegrown health products — an early-stage signal for novel, domestically sourced cannabinoid-adjacent therapeutics.
- A qualitative study found mothers reported medicinal cannabis as a facilitator of positive parenting, enabling them to manage anxiety, endometriosis and arthritis while maintaining caregiving capacity (Drug and Alcohol Review / Wiley).
🇯🇵 Japan — Health Ministry Shelves CBN Scheduling With No Implementation Date Set
🏛️ Regulation — CBN Sleep Aids Stay On Market As MHLW Defers Controlled-Substance Designation
Japan’s MHLW indefinitely postponed CBN’s designation as a controlled substance, with no implementation date set. CBN products — which have gained traction as sleep aids in Japan’s growing cannabinoid wellness sector — remain available in the market, though the shelved action signals continued regulatory scrutiny of non-THC cannabinoids.
🇮🇳 India — CSIR-IIIM Builds India’s First Medicinal Cannabis Facility In Jammu Under PPP
💼 Market — CSIR-IIIM Launches Jammu Medicinal Cannabis Facility Under PPP With French Partner
CSIR-Indian Institute of Integrative Medicine is developing India’s first cannabis-based medicinal project in Jammu under a public-private partnership with an unnamed French company, targeting export-quality painkillers for cancer and diabetic neuropathy. The facility has received Rs 55 crore in funding across ~8,000 sq m of built-up area; Union Minister Jitendra Singh confirmed the project has entered its third stage, with testing underway ahead of human trials. ⏳ Human trials pending.
🔬 Science — Northern India Cannabis Diversity And Dysmenorrhoea Formulation Trial
A peer-reviewed study mapped cannabinoid and terpene diversity in Cannabis sativa from Northern India, finding that cannabinoids and terpenes co-synthesise in capitate stalked and sessile glandular trichomes while bulbous glands produce only terpenes — establishing a foundational dataset for breeding strategies targeting India’s indigenous genetic resources.
- Hemp Street is conducting a pre/post-test clinical study at Amrita Vishwa Vidyapeetham evaluating the analgesic activity of Formulation Femme (Trailokya vijaya vati plus) in dysmenorrhoea management, with a sample of 60 participants.
- Innocan Pharma secured its first patent in India for a liposomal CBD injection targeting chronic pain — gaining IP protection in India’s US$55 billion pharmaceutical market (cross-ref Israel).
🇵🇰 Pakistan — Cabinet Approves Pakistan’s First National Cannabis Policy As Senate Streamlines Authority
🏛️ Regulation — Cabinet Approves First National Cannabis Policy As Senate Streamlines Authority Act
Pakistan’s federal cabinet approved the National Cannabis Control and Regulatory Policy 2025 under Section 13 of the Cannabis Control and Regulatory Authority Act 2024 — the country’s first national cannabis policy — enabling regulated cultivation and derivatives manufacturing for medicinal and industrial purposes. A high-level committee convened by the defence minister held two meetings (August and October 2025) before endorsing the framework with modifications; Prime Minister Shehbaz Sharif chaired the approving session.
- The Senate subsequently passed the Cannabis Control and Regulatory Authority (Amendment) Bill 2026, already approved by the National Assembly, realigning routine administrative powers from the federal cabinet to appropriate authorities. The amendment follows the Supreme Court‘s Mustafa Impex (PLD 2016 SC 808) judgment on collective cabinet decision-making, aiming to reduce procedural delays in the Cannabis Control and Regulatory Authority‘s operations.
🔬 Science — Hemp Cultivar Priming And Germination Establishment
A Scientific Reports study evaluated priming treatments on germination and seedling establishment of four hemp (Cannabis sativa L.) cultivars, addressing limited knowledge on seed priming techniques and growth parameters across soil and cocopeat media — relevant to Pakistan’s emerging cultivation framework where feminised seed costs of ~US$5–10 each make germination optimisation commercially significant.
🇰🇷 South Korea — CBD Reshapes Gut Microbiome To Promote Endurance In Mice
🔬 Science — Bifidobacterium Animalis And Oxidative Fibre Shift Underpin Gut–Muscle Axis Finding
A preclinical study found CBD reshapes the gut microbiome to promote endurance exercise in mice, increasing Bifidobacterium animalis abundance and oxidative muscle fibre composition after four weeks of treatment. Both CBD and B. animalis independently improved treadmill performance, suggesting a gut–muscle axis mechanism for cannabinoid-enhanced exercise capacity (Experimental & Molecular Medicine / Nature).
🇨🇳 China — CBD As Adjunctive Antitumour Agent Reviewed
🔬 Science — Systematic Review Compiles Multi-Cancer CBD Growth-Inhibition Mechanisms
A Harbin Medical University systematic review compiled preclinical evidence on CBD as a potential anti-tumour agent, highlighting mechanisms of tumour growth inhibition across multiple cancer models. Clinical validation remains pending; the authors position CBD as an adjunctive candidate warranting controlled trials (Phytomedicine).
🇦🇪 UAE — Cannabinoids In Neurological Conditions: 47-Trial Systematic Review
🔬 Science — Phase 2 Trials Dominate But Standardised Protocols Remain Absent
A systematic review identified 47 clinical trials investigating cannabinoids in neurological conditions, with multiple sclerosis, fibromyalgia and Parkinson’s disease the most frequently studied. Most trials were Phase 2, focused on pain, spasticity and cognitive function; the authors flag lack of standardised protocols, dosing and outcome measures as barriers to broader implementation (Frontiers in Pharmacology).
🇮🇷 Iran — Population Genomics Reveals Novel Cannabis Loci For Breeding
🔬 Science — Iranian Multi-Climate Collection Yields Loci For Flowering, Sex And Chemotype
A population genomics study of a natural Cannabis sativa collection from Iran — spanning male and female individuals from diverse climatic zones — identified novel genetic loci associated with flowering time, morphology, sex determination and chemotype (BMC Plant Biology). The findings expand the genomic toolkit available to breeders targeting climate-adapted, chemotype-specific cultivars.
🇱🇧 Lebanon — Cannabis Extract Protects Against Cisplatin Nephrotoxicity In Mice
🔬 Science — Lebanese Extract Inhibits Podocyte Apoptosis In Chemotherapy-Adjunct Model
A preclinical study found a Lebanese Cannabis sativa extract protected mice from cisplatin-induced nephrotoxicity by inhibiting podocyte apoptosis — suggesting a renoprotective mechanism relevant to chemotherapy adjunct therapy (Journal of Cannabis Research / BMC).
🇬🇭 Ghana — First West African State Launches Regulated Cannabis Programme
🏛️ Regulation — NACOC Launches Licences As Multinationals Line Up
Ghana‘s Interior Minister Muntaka Mohammed-Mubarak officially launched the Cannabis Regulatory Programme, establishing a controlled framework for low-THC hemp cultivation (≤0.3% THC) for medicinal and industrial purposes under the Narcotics Control Commission (Amendment) Act 2023. Referencing Canada‘s C$894.6 million cannabis-sector revenue, the minister positioned the programme to generate significant investment, rural employment and state revenue — making Ghana the first West African state to operationalise a regulated cannabis regime.
- The NACOC began issuing cultivation licences under an 11-category regime with a non-refundable $1,000 application fee and entry-level licence cost of $11,500 for 0.4 hectares. Licensing is restricted to Ghanaian citizens or permanent residents; corporate applicants must maintain at least 50% Ghanaian ownership.
- The Hempire Association of Ghana called the fees “the highest in the world,” arguing the tiered structure favours established entities over small farmers. The Chamber of Cannabis Industry received 1,000+ enquiries within six months — 60%+ from US and Canadian investors — and president Mark Darko forecast a $1 billion annual windfall for the local cannabis economy.
- The announcement caps a three-year wait since the Legal Instrument gave Act 1019 enforcement powers; long delays between legislative passage, fee drafting and licence issuance have estranged many aspiring stakeholders. NACOC will coordinate implementation with the Ministries of Interior and Agriculture, Ghana Standards Authority and Food and Drug Authority.
🇧🇼 Botswana — First Cannabis Licence Issued As Budget Allocation Confirmed
🏛️ Regulation — Swedish HIB Secures Dual GACP/EU-GMP Licence; P1.77 Billion Earmarked For Diversification
Botswana issued its first-ever cannabis licence in February, granting Swedish multinational Hemp Innovations Europe‘s local subsidiary HIB a dual licence for medical cannabis and industrial hemp cultivation under GACP and EU-GMP standards.
- The government views cannabis as an alternative forex source to diamonds; an MoU signed on 12 September 2025 between the BITC and HIE — with the Office of the President, BUAN and NARDI as parties — provides the institutional framework. Trade Ministry PS Joel Ramaphoi cited a “multiplier effect on job creation, investment, and industrial linkages.”
- Finance Minister Ndaba Gaolathe confirmed the policy shift in the 2026/2027 Budget Speech, allocating P1.77 billion (7.59% of the development budget) to agricultural diversification including cannabis. Implementation will be phased, beginning with trials at BUAN before expanding to farmers who demonstrate capacity. The budget explicitly provides for a Cannabis Production and Regulatory Framework.
- The Cannabis Association of Botswana urged phased licensing and citizen participation to avoid replicating South Africa‘s policy confusion. CAB member Sekgele Mogobe (Leaf Bantu) called for minimum citizen ownership thresholds, mandatory skills transfer, and tiered fee structures — warning that current export-oriented regulations risk favouring multinationals over local entrepreneurs.
- At the ‘Last Mile’ conference in Gaborone, SACHIDA‘s Cian McLelland said landrace varieties and Indigenous Knowledge Systems are central to local ownership of the cannabis value chain.
🇿🇼 Zimbabwe — $20,000 Licence Renewal Fee Squeezes Farmers As Exports Dwindle
🏛️ Regulation — Regulatory Costs Outpace Revenue Growth, Threatening Sector Sustainability
Zimbabwe’s cannabis export sector faces renewed uncertainty as a US$20,000 licence renewal fee looms over farmers already contending with dwindling exports and rising operational costs. The fee structure threatens to further compress margins in a market where regulatory costs have outpaced revenue growth, raising questions about the sustainability of the country’s licensed cannabis industry.
🇳🇦 Namibia — High Court Upholds 115 g Dealing Presumption, Rejecting Constitutional Challenge
🏛️ Regulation — Windhoek Finds “Nothing Compelling” — Contrasting South Africa’s 1995 Precedent
The Windhoek High Court rejected a constitutional challenge to the 115g cannabis dealing presumption under section 10 of the Abuse of Dependence-Producing Substances and Rehabilitation Centres Act (1971) in a judgment delivered on 30 January 2026.
- Deputy judge president Hannelie Prinsloo found “nothing compelling” to justify striking down the provision, after applicant Juvani Swartbooi — found with 241g of cannabis valued at ~N$2,500 and sentenced to 12 months — argued the presumption infringes the right to be presumed innocent and the right to a fair trial.
- A similar presumption in South Africa‘s Drugs and Drug Trafficking Act was struck down by the Constitutional Court in 1995 — a contrast that underscores Namibia’s more conservative judicial posture on cannabis reform.
🇲🇺 Mauritius — First Indian Ocean Country To Launch Government-Led Cannabis Law Review
🏛️ Regulation — Working Group Reviews Dangerous Drugs Act Against Canada, Uruguay And Germany Models
The National Anti-Drug Council (NADC) activated a technical working group in January 2026 to conduct a comprehensive review of all cannabis-related legislation — making Mauritius the first small island developing state in the Indian Ocean region to undertake a formal government-led cannabis policy review of this scope.
- The working group’s mandate covers a full review of the Dangerous Drugs Act (2000), which classifies cannabis as a Schedule I substance alongside heroin and cocaine with possession penalties of up to five years’ imprisonment. The group will assess international models — including decriminalisation (Portugal), medical-only frameworks (Thailand pre-2024) and regulated adult-use markets (Canada, Uruguay, Germany) — and deliver formal recommendations to government on a timeline not yet publicly specified.
- The initiative follows the 2024 general election, in which several parties included cannabis reform in their platforms. Prime Minister Navinchandra Ramgoolam, returned to office in November 2024, has signalled openness to evidence-based reform without endorsing a specific model — the NADC working group serving as a mechanism to build a policy evidence base while maintaining political flexibility.
- Mauritius’s position as a financial and services hub raises the prospect that reform could extend beyond decriminalisation to include provisions for cannabis-related investment, processing or export — a model pursued with varying degrees of success by Lesotho, Malawi and South Africa. The outcome could also influence policy discussions in neighbouring jurisdictions including Madagascar, Réunion and the Seychelles.