Czechia’s medical cannabis programme grew 46% in 2025—the strongest year since 2021. Full-year SÚKL data confirms the GP reform partially worked: prescribing doctors rose 28% to 305, flower grew 35% (233 → 314 kg), and the extract market more than doubled (30 → 76 kg). Domestic EU-GMP producers now anchor supply, while Czech infrastructure increasingly serves cross-border trade. From 1 January 2026, adults can cultivate up to 3 plants and hold 100 g at home—but the medical channel remains the primary regulated corridor.
Table of Contents
Czechia’s medical cannabis programme has grown consistently over the past five years, with issued volumes rising from tens of kilograms in 2019 to 390 kg in 2025. The trajectory reflects expanding clinical participation and a shift in product mix, as extracts now account for about 20% of issued volume.
In 2025, SÚKL reported 54,748 prescriptions, corresponding to roughly 440 kg prescribed. On an issued basis, pharmacies dispensed 390 kg (314 kg flower + 76 kg extracts), up from 267 kg in 2024 (+123 kg, +46%). The gap between prescribed and issued volume reflects timing, non-collection, partial dispensing, and substitution across products. Lukas Hurt, director of Magazine Konopí and a Czech market expert, explains: “The gap exists because not all prescribed cannabis is actually collected by patients. Some patients pass away, and some simply don’t manage to pick it up within two weeks, then they have to ask the doctor to issue it again because the prescription is valid for only two weeks.”
46% growth as domestic producers anchor supply and cross-border trade scales
Total issued volume rose from 267 kg in 2024 to 390 kg in 2025 (+46%). Within that, flower grew from 233 to 314 kg (+35%) and extracts from 30 to 76 kg (+153%). On an annual basis, prescribed volume (440 kg) exceeds issued volume (390 kg), implying roughly 86% of prescriptions are dispensed. The gap reflects the two-week prescription validity window, patient non-collection, and timing effects.
Dispensation Trends in Czechia

Czech-grown, EU-GMP certified flower saw the bulk of product launches in 2025. Lagom Pharmatech exports to Germany via Drapalin Pharmaceuticals GmbH and supplies the local market through Czech Medical Herbs. SensiQure runs its own line in Czech pharmacies and maintains German partnerships with nimbus health GmbH and Phyto Hemp. ELKOPLAST CZ, Ecogrow, and Zenplanto complete the domestic cultivation base. Czechia also hosts what Hurt calls a “world first”—a medical cannabis cultivation facility located directly within a hospital in Brno, serving several dozen inpatients.
Rua Bioscience (New Zealand) launched through Motagon in September 2025, while New Garden Pharma and CANNERALD announced prospective entries. Local distributors Czech Medical Herbs, Motagon, and Astrasana Holding AG now anchor supply chains, connecting domestic and international producers to a pharmacy network that grew from 127 to 163 outlets.
Bottlenecks elsewhere pushed Czech operators to pivot beyond production and distribution: import, reprocessing, and re-export are scaling fast. Over 2,000 kg were exported to Germany in the past year. Canadian trade data shows close to 10,000 kg imported from North America. The infrastructure built for domestic supply is now increasingly serving cross-border demand.
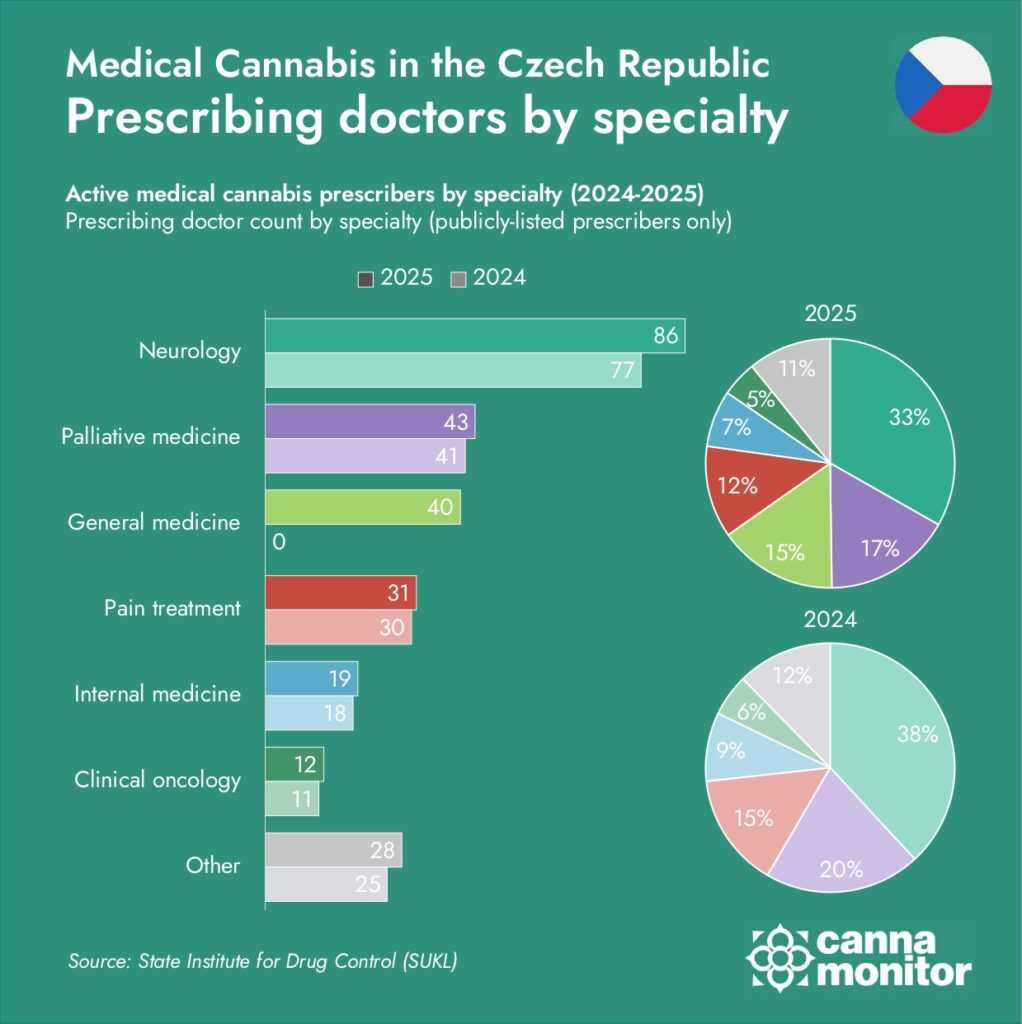
GP reform delivered 40 new prescribers—but the data overstates neurology
The April 2025 reform enabling GPs to prescribe for chronic pain expands the permission layer. The prescriber series reaching 305 by December (up 28% from 2024) marks the strongest growth since 2021. At least 40 new general medicine prescribers appeared in public data for the first time. The steepest monthly jump came between May and June (241 → 279 prescribers), right after implementation. Neurology still leads with 86 prescribers (33% share), but the base is diversifying.
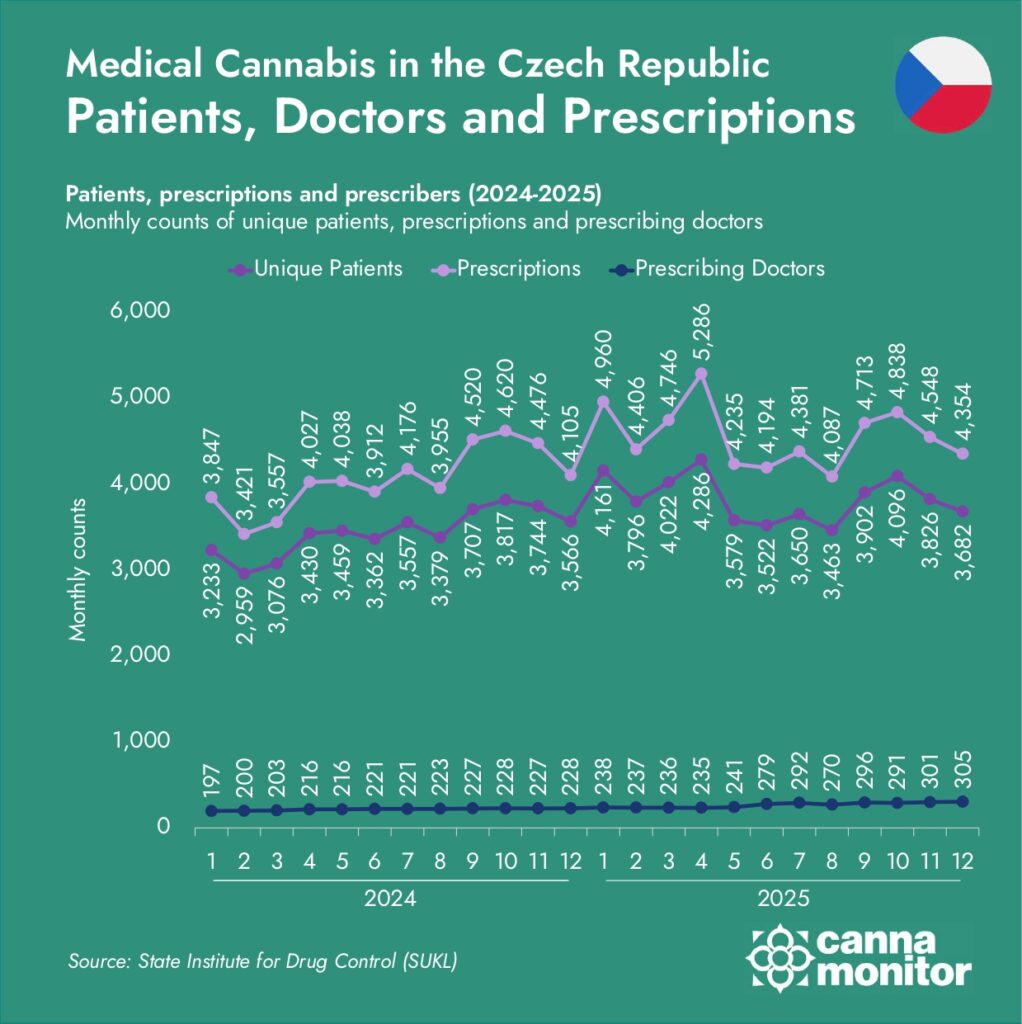
Three cautions help interpret these signals. First, SÚKL reports roughly 4,000 unique patients per month, but this understates the broader patient base. According to Hurt, “the total number of registered cannabis patients must be well over 10,000 today—some are not using it every month, some die, some stop for a certain period of time. Two years ago it was already above 8,000; last year it was close to 10,000.”
Second, SÚKL’s public prescriber directory lists doctors who agree to appear—not all active prescribers. Hurt notes that “many pain doctors didn’t agree to be listed there simply because they are already overwhelmed with existing cannabis patients and can’t accept more requests.” This selection bias likely overstates neurology’s share and understates pain specialists (algesiologists), who in practice dominate prescribing volume.
Third, chronic pain—not neurological conditions—drives most prescriptions. Hurt estimates chronic pain accounts for roughly 60–70% of the Czech market, with neurological disorders at 20–30% and other indications making up the remainder. This pattern mirrors most other medical cannabis markets.

Any Czech pharmacy can dispense medical cannabis if it chooses—there is no legal limit on outlets. Some sources cite 163 dispensing pharmacies, but Hurt clarifies this likely reflects pharmacies that have opted into public directories, not a regulatory cap. “Since 2021, any pharmacy in CZ can dispense medical cannabis if it wants to—like any other medicine,” he notes.
Healthcare figures for medical cannabis
Healthcare figures for medical cannabis
Decrim arrives in 2026 as Czechia cements its CBD and extract corridors
Czechia is moving on two tracks: criminal-law reform that reduces penalties and normalises limited personal cultivation, and the regulated medical pathway mediated through prescribers and pharmacy dispensing. From 1 January 2026, reforms allow adults to cultivate up to three plants and hold 100 g at home. This signals further normalisation, but it is not the creation of a commercial adult-use market. For patients who need consistent access, the medical channel remains the primary regulated corridor.
In parallel to medical scaling, Czechia has continued to attract CBD and low-THC activity. The country is emerging as a hub for CBD and hemp-derived intoxicating cannabinoids under the Psychomodulatory Substances Act framework, alongside continued uncertainty around EU Novel Food classification for CBD foods. Regulatory clarity for ≤1% THC is attracting businesses from Italy and Spain—and serving as an entry point for hemp-derived cannabinoid suppliers.
Within the medical channel, extracts are no longer marginal. Extracts grew from 30 kg in 2024 to 76 kg in 2025 (+153%), while flower grew from 233 kg to 314 kg (+35%). A larger extract segment makes the corridor look less like a flower-only pathway and more like a mixed-formulary pharmaceutical channel. That may change prescribing behaviour, patient adherence, and how product substitution shows up in the prescribed-versus-issued gap.
Beyond 2025: Can Czechia Lead Central Europe’s Cannabis Revolution?
Czechia’s medical cannabis programme matured further in 2025, with issued volumes up 46%, a measured gap between prescribed and issued volumes, rising prescriber participation, and a product mix that now includes a material extract segment.
The medical channel is showing scale potential: 390 kg dispensed to over 4,000 monthly patients (and an estimated 10,000+ cumulative patient base), 305 prescribers, a diversifying product mix, and the launch of the country’s first digital clinic.
The programme’s trajectory through 2026 will be shaped by two competing pulls: whether domestic demand scales enough to absorb growing supply—or whether operators increasingly pivot to international trading, leveraging Czech infrastructure as a reprocessing and re-export corridor for the wider European market.
This article draws on SÚKL data and expert input from Lukas Hurt, director of Magazine Konopí. On Tuesday 17 February at 3pm CET, Lukas and I will go deeper in a LinkedIn Live session covering the medical surge, the adult-use horizon, and Czechia’s positioning as a hemp gateway for the EU market.


